18 September 2020: Articles 
Syndrome of Inappropriate Antidiuretic Hormone (SIADH) Induced by Long-Term Use of Citalopram and Short-Term Use of Naproxen
Adverse events of drug therapy, Clinical situation which can not be reproduced for ethical reasons, Rare coexistence of disease or pathology
Chia-Yu Chiu1BCDEF*, Amara Sarwal1EF, Rehan Azhar Munir1AF, Michael Widjaja1CD, Amna Khalid1BC, Rajan Khanna1ADFDOI: 10.12659/AJCR.926561
Am J Case Rep 2020; 21:e926561
Abstract
BACKGROUND: Use of selective serotonin reuptake inhibitors (SSRIs) has been reported to be associated with the syndrome of inappropriate antidiuretic hormone (SIADH), although it is uncommon. Nonsteroidal anti-inflammatory drugs (NSAIDs), as a sole agent, are an even rarer cause of SIADH. Despite being documented in the literature, the understanding of the mechanism of both agents is limited. Here, we report a case of a patient taking both of these medications, a dangerous combination that led to the development of SIADH.
CASE REPORT: An 88-year-old woman with a history of asymptomatic chronic hyponatremia presented to our facility with symptomatic acute-on-chronic hyponatremia after she started using naproxen in addition to her daily citalopram. Her hyponatremia symptoms resolved after discontinuing these 2 offending agents, along with administration of fluid restriction and oral sodium supplements.
CONCLUSIONS: Naproxen is commonly prescribed and is often taken by elderly patients to control long-term or short-term pain. SSRIs, on the other hand, are a first-line treatment for depression and are usually prescribe by a psychiatrist. Hyponatremia is a rare medication adverse effect that should be kept in mind when treating these patients with either of these medications, and should especially be considered when combining them. Medication reconciliation should be done carefully by the provider to avoid adverse effects and drug interactions. When hyponatremia is encountered, options for future medication prescriptions include rechallenging with the same medication, switching to a different medication with the same mechanism of action, or using a medication from another class altogether. Monitoring of serum sodium level is warranted when titrating the offending agent.
Keywords: Anti-Inflammatory Agents, Non-Steroidal, Hyponatremia, Inappropriate ADH Syndrome, Serotonin Uptake Inhibitors, Aged, 80 and over, citalopram, naproxen
Background
In individuals with healthy kidneys, excess water intake can be excreted through diluted urine because of suppressed antidiuretic hormone. Syndrome of inappropriate antidiuretic hormone (SIADH) occurs via the loss of autoregulation of ADH, causing impaired water excretion. This leads to hyponatremia. A diagnosis of SIADH should be considered in euvolemic hyponatremic patients with urine osmolality >100 mOsm/kg, urine sodium >40 mmol/kg, and plasma osmolality <280 mOsm/kg. It is important to exclude differential diagnoses of hypothyroidism, glucocorticoid deficiency, hyperglycemia, hyperlipidemia and thiazide use [1]. Of note, there is no universal laboratory definition for SIADH. Measuring ADH level itself has not been used as a part of the diagnostic criteria in SIADH and is not warranted in routine practice.
Drug-induced SIADH can be simplified into 2 proposed mechanisms: (1) increased secretion of ADH or (2) increased renal response to ADH [2]. Serotonin reuptake inhibitors (SSRIs) work on the neurotransmitter norepinephrine. Under normal circumstances, norepinephrine stimulates the alpha 1 adrenergic receptor, which induces ADH release. Norepinephrine is then degraded and the signal it releases is terminated. When SSRIs are taken, norepinephrine reuptake is inhibited, and the signal continues to propagate. This causes increased stimulation of ADH release [2,3]. In contrast, nonsteroidal anti-inflammatory drugs (NSAIDs) appear to induce hyponatremia by potentiating the effects of ADH rather than by increasing ADH secretion. Normally, prostaglandin inhibits water reabsorption in the thick ascending limb and in the collecting tubules. NSAIDs inhibit renal prostaglandins synthesis, disturbing sodium and water homeostasis [2,4]. However, understanding of the mechanism involved is limited, and more studies are needed to elucidate the relationship between these 2 different classes of medications and their association with SIADH.
Case Report
An 88-year-old woman with hypertension, depression, osteo-arthritis over the bilateral shoulders, and bilateral knees presented to the Emergency Department (ED) because of generalized body ache. She also reported she had been experiencing nausea, decreased appetite, and malaise for 4 days.
Prior to admission, our patient walked with a cane indoors and used a walker when travelling outside. She was independent in her activities of daily living (ADLs) but was dependent on family support for instrumental activities of daily living (IADLs). She had been taking acetaminophen for osteoarthritis for more than 5 years, with no relief. Therefore, 2 weeks ago, her primary care physician decided to switch her pain medication from acetaminophen to naproxen.
Her reported home medications were amlodipine 10 mg daily, losartan 50 mg daily, citalopram 20 mg daily, and naproxen 250 mg every 8 h as needed. She reported she had been taking the naproxen 8 h every day for the past 2 weeks. Her medication list was verified by her pharmacy. She denied any other recent changes in medications, recent surgeries, vomiting, or diarrhea. She denied a history of heart failure, hepatic failure, hypothyroidism, adrenal insufficiency, malignancy, or HIV.
In the ED, her vital signs revealed a blood pressure of 169/82 mmHg, heart rate of 82 beats per minute, respiratory rate of 16 breaths per minute, and a temperature of 37.3°C. On exam, she was alert, awake, and oriented. Results of a neurological exam were unremarkable and she was able to walk about 10 meters with a cane. She was euvolemic and did not exhibit any peripheral edema or ascites. No other signs of heart failure or liver failure were noted. In addition, no goiter, excessively dry skin, brittle hair, or other physical findings consistent with hypothyroidism were found.
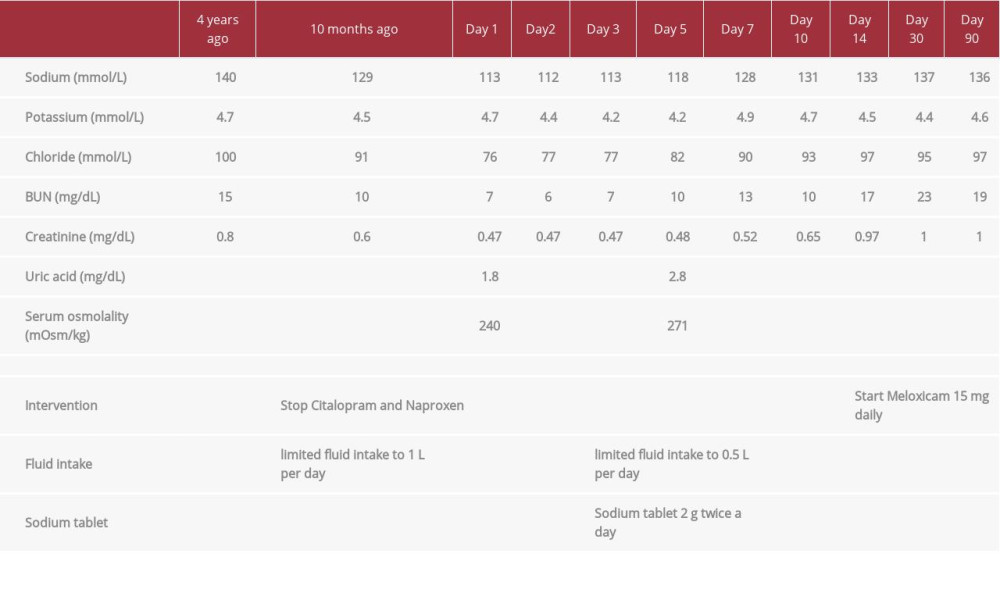
Laboratory testing revealed a fasting early morning cortisol level of 18 µ/dL, thyroid-stimulating hormone of 3.5 mU/L, pro-BNP of 221 pg/mL, creatinine 0.47 mg/dL, normal hepatic panel with a serum albumin of 4 g/dL, cholesterol 192 mg/dL, and triglyceride 44 mg/dL. Serum sodium was 113 mmol/L on admission, with a urine sodium of 120 mmol/L and urine osmolality 487 mOsm/kg. Her previous sodium level 10 months ago was 129 mmol/L (Table 1). Infectious causes were ruled out by imaging and microbiology. CT of neck, chest, and abdomen with contrast, along with brain MRI, were all unremarkable.
Based on our physical exam and laboratory testing, the differential diagnosis was SIADH. We stopped the offending agents, citalopram and naproxen, and started her on fluid restriction (fluid intake limited to 1 L per day). On Day 3, her serum sodium level remained at 113 mg/dL and she had worsening mental status. Although she was able to protect her airway, she became drowsy and was unable to follow commands. Our Endocrinology service recommended initiation of 2 g sodium chloride tablets twice a day as well as more aggressive fluid restriction to 0.5L per day. Her sodium gradually increased and was accompanied by improving mental status. Sodium chlo-ride tablets were stopped on Day 10 of admission, at which time the serum sodium was 131 mmol/L. We discharged our patient to a subacute rehabilitation center on Day 14. Her discharge medications included amlodipine 10 mg daily, losartan 100 mg daily, and meloxicam 15 mg daily for pain control. The need for SSRI or another antidepressant was assessed. The patient denied any depressive symptoms and was cleared by our inpatient psychiatry team. As hyponatremia on its own can induce psychological symptoms, we held the SSRI and advised her to follow up with an outpatient psychiatrist. Our patient and her family were instructed to limit fluid intake to 1 L per day.
She followed up in the Endocrinology Clinic on Day 30, which revealed an improvement in serum sodium to 137 mmol/L (Table 1). She was able to walk in the clinic with her cane and had no further complaints of nausea, decreased appetite, or malaise. She reported that the pain over bilateral knees and bilateral shoulder joints was improving. A comparison of laboratory data is displayed in Table 1. As her serum sodium was within normal range. A re-evaluation by Psychiatry was done and it was decided to stop the antidepressant.
Instead of using hypertonic saline in our case, we decided to start fluid restriction alone at the beginning because of advanced age (which entails higher risk for osmotic demyelination syndrome), chronic hyponatremia, and relatively stable clinical condition. The culprits of SIADH, which were citalopram and naproxen, were stopped promptly when the patient arrived at the ED. However, her hyponatremia failed to respond to fluid restriction and she developed worsening neurological symptoms. We decided to start her on oral sodium supplement 2 g twice daily with increasing restriction on fluids. After starting this treatment regimen, we saw an adequate improvement in her sodium level.
Discussion
SIADH is known to be associated with use of certain antidepressants. The incidence of SIADH in patients taking SSRIs is variable, ranging from 0.5% to 32% [3]. A retrospective study in Denmark reviewed around 68 000 patients who were using antidepressants [5], showing that hyponatremia was most strongly associated with use of citalopram (SSRI) compared with duloxetine (serotonin and norepinephrine reuptake inhibitor, SNRI), venlafaxine (SNRI), and mirtazapine (noradrenergic and specific serotonergic antidepressant, NaSSA). A similar result was found in a recent study showing that patients taking SSRIs were more likely to have hyponatremia compared to patients who took antidepressants that had other mechanisms of action. This effect is higher in the elderly, in females, when combined with diuretic use, in those with low body weight, and in those with baseline low sodium serum level [3,6,7]. It has been documented that hyponatremia due to SSRI can develop within the first few weeks to few months after initiation of treatment [6]. The mechanism of hyponatremia has been thought to be due to inhibition of norepinephrine reup-take [2,3]. However, this cannot fully explain why SSRIs are associated with a higher incidence of SIADH when compared with SNRIs or NaSSAs.
A limitation of the studies cited above is that the diagnosis of SIADH was not made in every patient, and not all individuals received the necessary workup, and this may have resulted in the overestimation of SIADH cases [5–7]. Mild hyponatremia itself (<130 mmol/L) without any symptoms may be ignored by clinicians and not investigated. In recent studies, it was found that by treating even mild hyponatremia and curing any reversible cause of SIADH, there is the potential to improve quality of life [8–10]. The practice of continuing these offending agents in asymptomatic mildly hyponatremic patient should be re-evaluated.
When discontinuing SSRIs, the amount of time taken to return to a normal serum sodium level can range from 48 h to 6 weeks, with most cases resolving within 2 weeks [3]. Not all of the cases required aggressive treatment. Some studies found that patients re-experience hyponatremia by restarting the antidepressants that initially gave them hyponatremia, although it was not common [3]. As a strict guideline does not exist about SSRI-induced SIADH, providers often rely on their own clinical experience. This can include another trial of the same antidepressant, switching to a different medication within the same class, or using an alternative medication from a different class.
NSAIDs is also a class of medications that can cause SIADH [1,2,4,11]. A 2013 review article showed at least 11 patients reporting SIADH secondary to NSAIDs were using ibuprofen (4/11) or diclofenac (2/11) [1]. A case report from Turkey discussed a 78-year-old man taking meloxicam 15 mg twice a day for 5 days who developed hyponatremia of 114 mmol/L [4]. Similar to the situation with SSRIs, there is currently not a guideline for choosing, switching, or rechallenging of NSAIDs in SIADH patients due to the paucity of data.
Conclusions
In our case, laboratory evaluation led to the diagnosis of SIADH, and the history of presenting illness pointed towards a medication adverse effect as the culprit. After ceasing the medication and providing appropriate treatment, our patient’s hyponatremia resolved. Currently, there is not a universal standardized protocol to guide clinicians when patients experience SSRI-induced SIADH or NSAID-induced SIADH. However, hyponatremia in the elderly is associated with neurocognitive decline, gait instability, falls, increased hospital readmission rate, increased need of long-term care, and increased mortality. When faced with hyponatremia in an elderly patient, medication reconciliation should be one of the initials steps in the assessment.
References:
1.. Artom N, Oddo S, Pende A, Syndrome of inappropriate antidiuretic hormone secretion and ibuprofen, a rare association to be considered: Role of tolvaptan: Case Rep Endocrinol, 2013; 2013; 818259
2.. Liamis G, Milionis H, Elisaf M, A review of drug-induced hyponatremia: Am J Kidney Dis, 2008; 52(1); 144-53
3.. Jacob S, Spinler SA, Hyponatremia associated with selective serotonin reuptake inhibitors in older adults: Ann Pharmacother, 2006; 40(9); 1618-22
4.. Demir ME, Horoz M, Ulas T, Nonsteroidal anti-inflammatory drug-induced severe hyponatremia: Medicina (Kaunas), 2012; 48(12); 619-21
5.. Leth-Møller KB, Hansen AH, Torstensson M, Antidepressants and the risk of hyponatremia: A Danish register-based population study: BMJ Open, 2016; 6(5); e011200
6.. Liu BA, Mittmann N, Knowles SR, Hyponatremia and the syndrome of inappropriate secretion of antidiuretic hormone associated with the use of selective serotonin reuptake inhibitors: A review of spontaneous reports: CMAJ, 1996; 155(5); 519-27
7.. Gandhi S, Shariff SZ, Al-Jaishi A, Second-generation antidepressants and hyponatremia risk: A population-based cohort study of older adults: Am J Kidney Dis, 2017; 69(1); 87-96
8.. Schrier RW, Gross P, Gheorghiade M, Tolvaptan, a selective oral vasopressin V2-receptor antagonist, for hyponatremia: N Engl J Med, 2006; 355(20); 2099-112
9.. Renneboog B, Musch W, Vandemergel X, Mild chronic hyponatremia is associated with falls, unsteadiness, and attention deficits: Am J Med, 2006; 119(1); 71.e1-8
10.. Koch CA, Fulop T, Clinical aspects of changes in water and sodium homeostasis in the elderly: Rev Endocr Metab Disord, 2017; 18(1); 49-66
11.. Roche C, Ragot C, Moalic JL, Ibuprofen can induce syndrome of inappropriate diuresis in healthy young patients: Case Rep Med, 2013; 2013; 167267
In Press
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942770
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943214
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943010
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943687
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250