22 February 2021: Articles 
Solitary Neurofibroma of the Hard Palate: A Case Report and Literature Review
Rare disease
Takaharu Taketomi12ABCDEF*, Ken Nakamura32BC, Yui Teratani1AB, Katsuhisa Matsuo1B, Jingo Kusukawa1DDOI: 10.12659/AJCR.929674
Am J Case Rep 2021; 22:e929674
Abstract
BACKGROUND: Neurofibromas are benign tumors of neurological origin caused by the proliferation of Schwann cells and fibroblasts; they often occur in the skin and nerves as a symptom of von Recklinghausen disease. Solitary neurofibromas are also known to occur on their own, but solitary development in the hard palate is extremely rare and difficult to distinguish from schwannomas. The neural origin of solitary neurofibromas is also difficult to determine intraoperatively, and there have been no reports that clearly identify the neural origin of neurofibromas in the hard palate.
CASE REPORT: We report a case of a solitary neurofibroma originating in the hard palate in a 24-year-old woman. She presented to our department with a 1.2×0.8-cm dome-shaped left palate mass. After identification of the nerve at the source, the tumor was resected under general anesthesia. Histopathology was positive for S-100 and CD34 immunostaining, as well as for Alcian blue. Eventually, the mass was diagnosed as a neurofibroma.
CONCLUSIONS: Solitary neurofibromas originating in the hard palate are difficult to differentiate from other neoplastic lesions, especially schwannomas, based on clinical findings alone. Therefore, it is important to perform a biopsy and immunostaining of the biopsied specimens for S-100 and CD34. In neurofibromas, tumor cells are loose and delicate, often with wavy or serpentine nuclei, and S-100 protein-positive cells are sparser than in schwannomas. An overall pathological diagnosis should be made with regard to CD34, taking into account that schwannomas are CD34-negative and neurofibromas are CD34-positive.
Keywords: Neurofibroma, Palate, Hard, S100 Proteins, Schwann Cells, Biopsy, Neurilemmoma, Neurofibromatosis 1, young adult
Background
According to the 2017 World Health Organization Classification of Head and Neck Tumors, neurofibromas are benign peripheral nerve sheath tumors that consist of a mixture of Schwann cells, perineurial cells, fibroblasts, and axons [1]. Neurofibromas resemble schwannomas and are often seen as elastic, hard, well-defined masses with no capsule. The World Health Organization has classified neurofibromas into 2 types: dermal and plexi-form [2]. Dermal neurofibromas arise from a single peripheral nerve, while plexiform neurofibromas are associated with multiple nerve bundles.
Neurofibromas may appear as part of neurofibromatosis type I (NF-1) or von Recklinghausen disease, and they present most frequently with café-au-lait spots on the skin [3]. Solitary neurofibromas rarely occur in the oral cavity [4]. The frequency of solitary neurofibromas in the oral cavity is reportedly 6.5%, particularly for lesions that are not associated with NF-1 [5], and their occurrence on the hard palate is extremely rare.
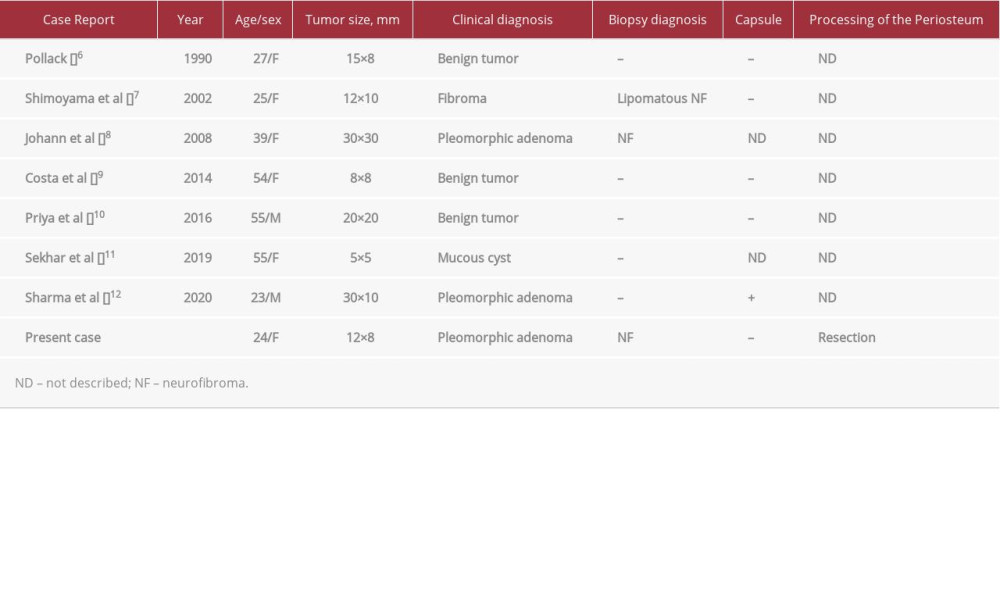
There have only been 7 published cases of solitary neurofibroma of the hard palate (Table 1) [6–12]. The mean patient age in these cases was 40±15 years, and there were 5 women (71%), suggesting a higher incidence among women. The clinical diagnoses were as follows: benign tumor in 3 cases (43%); pleomorphic adenoma in 3 cases (43%); and mucous cyst in 1 case (14%). Biopsies were performed in 2 cases (29%), and a neurofibroma was diagnosed in 1 case (14%).
Herein, we report a case of solitary neurofibroma occurring in the hard palate that originated in the greater palatine nerve.
Case Report
LOCAL FINDINGS AND TREATMENT:
On physical examination, the patient was well-nourished and had no abnormalities on her body or facial skin. We observed a well-defined dome-shaped mass measuring 1.2×0.8 cm on the hard palate mucosa adjacent to the left upper molars (Figure 1A). The mass had a smooth surface and a normal mucosal color. The elasticity was slightly hard, and no waves were palpable. There was no spontaneous pain or tenderness, and no palatal hypoesthesia was noted. The patient did not experience any spontaneous pain, tenderness, or palatal hypesthesia.
In the radiographic examination, we did not observe any abnormal bone resorption around the tumor on plain radiographs of the maxillary occlusion (Figure 1B) or on computed tomography images (Figure 1C). T2-weighted magnetic resonance imaging revealed a tumorous lesion with hyperintensity on the left posterior hard palate (Figure 2A, 2B).
The clinical diagnosis was a pleomorphic adenoma of minor salivary gland origin, and a preoperative biopsy was performed to confirm the diagnosis. The pathology differed from the clinical diagnosis, however; it revealed a nervous system tumor rather than a pleomorphic adenoma. Therefore, we performed a tumor resection while the patient was under general anesthesia. We demarcated a resection line with a 0.2-cm margin from the base of the broad-based tumor and resected the superficial layer, comprising the mucous membrane and the deep part comprising the periosteum (Figure 3A–3D). We also cut and ligated the nerve and vascular bundles, which appeared to be branches of the large palatal nerve connected to the tumor (Figure 3B, 3C). After resection, the bone surface was smooth, and there was no abnormal bone resorption (Figure 3D). We completed the surgery by covering the area of resection with an artificial dermis (TERDARMIS) and attaching a protective floor. The size of the resected specimen was 1.8×1.2×1.0 cm, and it had a round, smooth surface and slightly soft elasticity (Figure 3E, 3F).
HISTOPATHOLOGICAL FINDINGS:
Histopathological findings showed that the tumor parenchyma was located in the submucosa, with relatively well-defined borders but without a clear fibrous capsule (Figure 4A). Small cells (Schwann cells and fibroblasts) with corrugated or spindle-shaped nuclei and fibrous components were present in specimen sections stained with hematoxylin and eosin (Figure 4B). Immunostaining for S-100 protein localized in the cytoplasm and nucleus of nervous system cells showed that S-100 protein-positive cells were scattered and present in a wavy arrangement (Figure 4C).
In 1994, Chaubal et al [13] reported CD34-positive spindle cells in neurofibromas; CD34, which localizes to the cell membrane and cytoplasm, is found in neurofibromas but not schwannomas. In our patient, immunostaining of the specimen revealed spindle-shaped cells that were positive for CD34 (Figure 4D). Furthermore, staining with Alcian blue was positive, suggesting mucous stromal tissue (Figure 4E). On histopathology, neurofibromas have a myxomatous stroma, whereas schwannomas do not [11].
There was no capsule in this tumor, the tumor cells were arranged in a wavy pattern, S-100 protein-positive cells were scattered, and CD34-positive cells and myxoma-like stroma were found. Based on these features, the final diagnosis was a neurofibroma. Five years have passed since the surgery, and the patient has not experienced any recurrences.
Discussion
Neurofibromas are benign lesions of the peripheral nervous system derived from Schwann and mesenchymal cells of the nerve sheath. Neurofibromas can appear as single tumors (solitary neurofibromas) or as NF-1 that presents as multiple tumors along with café-au-lait spots in von Recklinghausen disease. Neurofibromas are common in the tongue and buccal mucosa in the oral cavity, but approximately 20–60% of oral neurofibromas are associated with neurofibromatosis [14]. In 2019, Broly et al [15] reviewed 26 cases of isolated neurofibromas occurring in the oral cavity [15], and intraoral occurrences were very rare. However, because the oral cavity is composed of various tissues such as the tongue, buccal mucosa, gingiva, floor, and palate, the neurofibromas that develop there have different nerves of origin and different surrounding tissues that are removed during surgery. Therefore, we focus here on a solitary neurofibroma arising in the hard palate, along with a review of the literature reported through 2020.
Solitary neurofibromas of the hard palate, as in the present case, are extremely rare, and only 7 cases have been reported to date [6–12]. In general, solitary neurofibromas are difficult to differentiate from schwannomas. A biopsy was also obtained in this case, and the mass was diagnosed as a neurogenic tumor, such as a schwannoma or neurofibromatosis, but it was difficult to distinguish the type.
In general, schwannomas are classified as Antoni A, Antoni B, or a mixture of the 2 types, with Antoni A being characterized by a fenestrated or ornithological arrangement of the nuclei and the presence of Verocay bodies. In Antoni B, the tumor cells are sparsely arranged, and the stroma shows cyst formation and mucus-like degeneration. Usually, either type is rarely seen on its own, and often a mixture of both types is observed. Neurofibromas, on the other hand, have spindle-shaped tumor cells that are less cytoplasmic than schwannomas, and their nuclei are small, oval in shape, and do not exhibit a palisade pattern. Because of the fibrous components found between the cells, the tumor cells appear wavy [1,11]. In addition to this arrangement of cells, S-100 protein positivity is a major differentiator between neurofibromas and schwannomas, with neurofibromas being positive for S-100 protein, but at a lower rate than schwannomas [1]. Chaubal et al [13] and Ohno et al [16] reported that CD34-positive spindle-shaped cells are found in neurofibromas but not in schwannomas. On histopathology, the myxomatous stroma is seen in neurofibromas, but not in schwannomas [11]. In addition, schwannomas often have capsules, while neurofibromas do not [17]; therefore, neurofibromas have been reported to be more likely to recur than schwannomas [18]. In previously reported cases, no capsules were found in 4 of 7 patients (57%). However, there have been reports of capsules in neurofibromas [1], and we believe that the presence or absence of capsules is not a characteristic that differentiates the 2 tumors. In the case of a tumor without a capsule, the setting of the resection margin is very difficult. On the hard palate, the mucosa is present on the surface, and the periosteum is present in the deeper layers; therefore, if these are included in the resection, vertical margins do not present a problem. Alternatively, surgeons should be careful with the horizontal margins because there is often no capsule. The specific numerical values of the margin’s widths are unknown. Here, we set the horizontal margin to 0.2 cm and excised the palatal mucosa (surface layer) and periosteum (deep layer), which made complete resection possible.
There were no recurrences in any of the 7 previously reported cases or in our case. Recurrence can be avoided by providing a safety margin, including the periosteum, around the tumor at the point of resection. Here, the tumor, which arose in the hard palate, was attached to a greater palatal nerve at the time of resection. Although there is no indication of whether the nerves were identified in the other 7 cases [6–12], they were probably derived from the greater palatal nerve based on the tumor’s location.
A neurofibroma of the hard palate needs to be differentiated from many other diseases, especially salivary gland tumors. The S-100 protein is a useful marker for identifying nervous system tumors, and it is important to perform a preoperative biopsy. If the histopathological specimen at the time of biopsy is positive for S-100 protein, a benign neurogenic tumor is likely. In this case, schwannoma and neurofibroma were possible candidates. After differentiation by cell alignment style, S-100 protein distribution, and CD34 staining, vertical margins should include the mucosa (superficial) and periosteum (deep), and horizontal margins should be excised with a safety margin, considering the absence of capsules. Regarding the malignant transformation, a solitary neurofibroma is considered less likely to become malignant compared with neurofibromatosis [19], but sufficient future follow-ups are necessary to track its course.
Conclusions
To the best of our knowledge, the present case is the 8th reported case of a neurofibroma arising in the hard palate and the first to specifically identify the nerve of origin (the greater palatal nerve) by surgical photography. Preoperative biopsy and immunohistochemical analysis are important. Adequate margins should be established at the time of resection because the tumor often has no membrane. In this case, a deep periosteal resection was performed and the horizontal margin was set at 0.2 cm. Five years have passed since the surgery, and the patient has not developed any recurrences to date.
Figures
References:
1.. El-Naggar AK, Chan JKC, Grandis JR: World Health Organization classification of head and neck tumors, 2017; 123-24, Lyon, IARC Press
2.. Mahalle A, Gs Reddy M, Mohit Kheur S, Solitary non syndromic oral plexiform neurofibroma: A case report and review of literature: J Dent (Shiraz), 2016; 17; 293-96
3.. Geist JR, Gander DL, Stefanac SJ, Oral manifestations of neurofibromatosis types I and II: Oral Surg Oral Med Oral Pathol, 1992; 73; 376-82
4.. Depprich R, Singh DD, Reinecke P, Solitary submucous neurofibroma of the mandible: Review of the literature and report of a rare case: Head Face Med, 2009; 5; 24
5.. Surayama S, Shashikumar P, Shreeshyla HS, Sheela Kumar G, Solitary plexi-form neurofibroma of the gingiva: Unique presentation in the oral cavity: J Clin Diagn Res, 2013; 7; 2090-92
6.. Pollack RP, Neurofibroma of the palatal mucosa. A case report: J Periodontol, 1990; 61; 456-58
7.. Shimoyama T, Kato T, Nasu D, Solitary neurofibroma of the oral mucosa: A previously undescribed variant of neurofibroma: J Oral Sci, 2002; 44; 59-63
8.. Johann AC, Caldeira PC, Souto GR, Extra-osseous solitary hard palate neurofibroma: Braz J Otorhinolaryngol, 2008; 74; 317
9.. Costa FW, Carvalho FS, Sousa CF, Solitary neurofibroma of the palate: Braz J Otorhinolaryngol, 2014; 80; 184-85
10.. Priya M, Bakshi S, Coumare V, Solitary extraosseous neurofibroma of hard palate: Report of a case with a review of literature: J Dent Allied Sci, 2016; 5; 95-97
11.. Sekhar P, Nandhini G, Kumar KR, Kumar AR, Solitary neurofibroma of the palate mimicking mucocele: A rare case report: J Oral Maxillofac Pathol, 2019; 23; 23-26
12.. Sharma P, Kamboj M, Narwal A, Palatal solitary neurofibroma simulates a salivary gland neoplasm: J Oral Maxillofac Surg Med Pathol, 2020; 32; 76-78
13.. Chaubal A, Paetau A, Zoltick P, Miettinen M, CD34 immunoreactivity in nervous system tumors: Acta Neuropathol, 1994; 88; 454-58
14.. Wright BA, Jackson D, Neural tumors of the oral cavity. A review of the spectrum of benign and malignant oral tumors of the oral cavity and jaws: Oral Surg Oral Med Oral Pathol, 1980; 49; 509-22
15.. Broly E, Lefevre B, Zachar D, Hafian H, Solitary neurofibroma of the floor of the mouth: Rare localization at lingual nerve with intraoral excision: BMC Oral Health, 2019; 19; 197
16.. Ohno J, Iwahashi T, Ozasa R, Solitary neurofibroma of the gingiva with prominent differentiation of Meissner bodies: A case report: Diagn Pathol, 2010; 5; 61
17.. Zachariades N, Mezitis M, Vairaktaris E, Benign neurogenic tumors of the oral cavity: Int J Oral Maxillofac Surg, 1987; 16; 70-76
18.. Gnepp DR, Keyes GG, Central neurofibromas of the mandible: Report of two cases: J Oral Surg, 1981; 39; 125-27
19.. Abell MR, Hart WR, Olson JR, Tumors of the peripheral nervous system: Hum Pathol, 1970; 1; 503-51
Figures
In Press
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943687
17 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943070
17 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943370
18 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943803
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250