18 August 2021: Articles 
A 2-Year-Old Boy Who Developed an Aortoesophageal Fistula After Swallowing a Button Battery, Managed Using a Novel Procedure with Vascular Plug Device as a Bridge to Definitive Surgical Repair
Management of emergency care, Rare disease, Educational Purpose (only if useful for a systematic review or synthesis)
Khalid M. Alreheili1BCDEF*, Mansour Almutairi2EF, Ali Alsaadi3EF, Ghousia Ahmed4F, Abdulrahman Alhejili4F, Taha AlKhatrawi5BFDOI: 10.12659/AJCR.931013
Am J Case Rep 2021; 22:e931013
Abstract
BACKGROUND: Button batteries (BBs) can be inhaled or swallowed accidentally, particularly by infants and children, who can present as a surgical emergency with a fatal outcome. This report is of a case of a 2-year-old boy who developed an aortoesophageal fistula (AEF) after swallowing a button battery and was successfully treated using a novel vascular plug device as a bridge to definitive surgical repair. AEF is diagnosed using computed tomography angiography (CTA), after laparotomy, and using aortography. Aortic endovascular stenting and vascular occluder placement is a minimally invasive emergency option until definitive treatment can be provided. The surgical options include repair the aortic defect primarily, or the diseased segment of the aorta is replaced with a graft.
CASE REPORT: A 2-year-old boy presented with hematemesis 9 days after ingestion of a button battery, which was removed endoscopically 16 hours after the incident. The patient was resuscitated aggressively and diagnosed to have AEF using computed tomography angiography (CTA). The aorta was successfully repaired using a vascular plug device, which kept the patient safe until the definitive surgical treatment was done 2 months later. The defect was repaired with direct anastomosis and completed with a patch of bovine pericardium, as well as closure of the fistula from the esophageal side with stitches.
CONCLUSIONS: This report presents a rare but previously reported complication of swallowing a button battery, resulting in an aortoesophageal fistula. The aorta was successfully repaired using a vascular plug as a minimally invasive emergency option, which is considered as a lifesaving procedure and a bridge for definitive repair.
Keywords: Fatal Outcome, Foreign Bodies, Pediatrics, Survival, Vascular Fistula, Aortic Diseases, Blood Vessel Prosthesis Implantation, Child, Preschool, Deglutition, Esophageal Fistula
Background
Foreign body (FB) ingestion is common among young children and is mostly an uneventful incident. Complications resulting from FB ingestion depend on the nature of the FB, size of the FB, site of dislodgement, duration of retention of FB, presence of any anatomical abnormalities, and the age of the child. Button batteries (BBs) are among the most dangerous objects that are commonly ingested, especially in the esophagus, and their removal requires emergency endoscopy [1]. Many serious complications have been reported with BB ingestion. These include perforation, mediastinitis, gastrointestinal (GI) bleeding, esophageal stricture, tracheoesophageal fistula, and aortoesophageal fistula (AEF) [2]. AEF was fatal in most reported cases. If AEF is suspected, CTA is the preferred initial diagnostic test. The management of AEF in children includes aggressive resuscitation, measures to control bleeding (eg, use of balloons), procedures like percutaneous endovascular aortic repair (EVAR), or our novel use of a vascular plug device to close the fistula as a bridge to definitive surgical repair.
Case Report
An otherwise healthy boy, age 2 year 6 months, presented to the Emergency Department on 17 May 2020 at 9 PM with a history of frequent vomiting and difficulty in swallowing semi-solid food. Accidental battery ingestion was suspected when his family noticed the absence of a battery in the glucometer device at 5 AM. The estimated time since the ingestion was 16 h. On physical examination, the patient generally looked well; he was not tachypneic or distressed. He was mildly dehydrated and showed stable vital signs, and the results of the systemic examination were unremarkable. Chest radiography revealed a BB located in the upper part of the esophagus. The patient was admitted to the hospital and was kept nil per os (NPO). An urgent upper gastrointestinal endoscopy (EGD) was performed under general anesthesia within 2 h of his arrival to the hospital, and the endoscope inserted to 15 cm. A disc battery was found, with a circumferential area of necrosis. The battery was fixed to the mucosa and it was removed using rat-tooth forceps. The area was found to have circumferential third-degree burns at 15 cm (Figure 1). The patient stayed in the hospital for 5 days. On the first 2 days, he was kept NPO and was administered intravenous omeprazole. Chest radiography was repeated, with normal results, and no contrast study (esophagogram) done. During the next 2 days, intake of only water was allowed, following which intake of other liquids was permitted. The patient tolerated feeding and was discharged in good condition on the fifth day. On 26 May 2020, almost 4 days after discharge, the patient presented to our Emergency Department at 9 PM with a history of melena for 1 day, and nasal bleeding and hematemesis on the day of admission. The mother gave a history of cough, runny nose, and fever since the last 2 days. On examination, the patient looked generally unwell, febrile, extremely pale, tachypneic, and dehydrated. His vital signs were as follows: temperature 38.3°C, heart rate (HR) 175 beats per min, respiratory rate (RR) 30 breaths per min, BP 72/44 mmHg, SpO2 100% on face mask 10 L/min, blood gas PH: 7.08, PCo2 42, HCO3 11.2, lactate 7.9, and random blood sugar 278 mg/dl (possibly due to stress, as it later normalized). Cardiovascular examination revealed normal first and second heart sounds and hemic murmur; no gallop was heard. Examination of the respiratory system revealed equal bilateral air entry, with no added sounds. Gastrointestinal examination showed a mildly distended but soft and lax abdomen with no organomegaly. In the emergency room, normal saline boluses of a total 40 ml/kg were administered, as well as packed red blood cells (PRBCs) 10 ml/kg, tranexamic acid stat, omeprazole, and paracetamol, as he was febrile. An urgent computed tomography (CT) with contrast was performed after he was sedated with 2 doses of midazolam and lorazepam. CT angiography (CTA) of the chest and abdomen showed a fistula communication between the aorta and esophagus at the level of the T4 vertebra distal to the left subclavian artery. The fistula measured approximately 3.3 mm in transverse diameter, with contrast media filling the dilated esophagus and extending to the stomach, suggestive of active bleeding (Figure 2). A suspicious fistula was noted between the trachea and esophagus at the level of the right main bronchus. The patient was shifted to the pediatric intensive care unit (PICU) after resuscitation with 2 boluses of normal saline at 20 ml/kg. His BP was 86/45 mmHg, HR 140 bpm, and he was tachypneic. The patient received PRBCs a second time immediately in the PICU. Elective rapid sequence intubation was performed. The interventional cardiologist was ready to intervene and close the fistula from the aortic side using the device. Therefore, an urgent referral was made to the nearby cardiac center (10 to 15 minutes away from our hospital). In the cardiac center, the patient received 1 unit of PRBCs attached, a central line was inserted in the internal jugular vein under ultrasonography (US), and complete aseptic technique was used. Another unit of PRBCs and 1 unit of platelets (PLT) and fresh frozen plasma (FFP) were administered owing to massive bleeding during the procedure. The patient was administered sodium bicarbonate twice for acidosis. The procedure was performed under general anesthesia and intubation. The right femoral artery was punctured, and a 5F sheath was introduced using the Seldinger technique. A 4F JR diagnostic catheter (JR 3.5, 100 cm×0.038”, Cordis Company, Florida, USA) was introduced into the descending aorta, and an aortogram was obtained using hand injection contrast. A selective fistulogram was performed to delineate the course of the fistula. A guidewire (radiofocus guidewire, 0.035×150, Terumo Corporation, Japan) was used to cross the fistula and was placed inside the esophagus. Thereafter, a 5F-long delivery system was introduced in exchange with a short sheath. The Amplatzer Duct Occluder II 6×6 mm (ADO II; St. Jude Medical, St. Paul, MN, USA) was used to occlude the fistula (Figure 3). The device position was checked using an aortogram from the side arm of the long delivery sheath. The device was released in a good position, and an aortogram was obtained at the end, which showed no more leakage. At the end of the procedure, all catheters and wires were removed, and manual compression was used to stop bleeding from the punctured side. The patient remained well sedated in the PICU on fentanyl 4 μg/kg/h and midazolam 4 μg/kg/min, ventilated on SIMV pressure control. The NPO was maintained, and NGT was inserted by a pediatric surgeon to decompress the distended stomach, and the patient was started on total parenteral nutrition. As the patient still had fever, ceftriaxone was discontinued, and meropenem plus vancomycin was started. A chest CTA, obtained on the fifth day, showed interval placement of the vascular plug through the previously seen AEF. Currently, it is seen extending between the aorta and esophageal lumen with no active extravasation (Figure 4). The esophagus remained dilated with a persistent fistula noted between the trachea and the esophagus at the level of the right main bronchus (esophageal-bronchial fistula. A plain CT scan of the brain showed a well-defined hypodensity involving the right occipital lobe with surround mass effect and effacement of the cortical sulci, denoting brain infarction involving the right PCA territory. Echocardiography was normal. The patient was extubated after 1 week and was stable with only melena in the first few days, although without a significant drop in hemoglobin levels, as the patient pulled out the NGT and he was therefore continued on total parenteral nutrition (TPN) the next week. An upper GI contrast study revealed fistula communication between the esophagus and the right main bronchus. The esophageal stricture could not be fully assessed as a small amount of contrast was given to avoid aspiration, so laparoscopic gastrostomy tube insertion was performed. Enteral feeding was advanced, and TPN was weaned. The patient was discharged in the fourth week. CTA was repeated in the sixth week after device insertion, and shows closure of the tracheoesophageal fistula and the device in place. The patient was referred to a higher-level care center for an opinion regarding the long-term plan for this patient. As the long-term plan for this device is unknown for them, cardiothoracic surgeons prefer to remove this device electively, and they repair the defect posteriorly with direct anastomosis between the 2 ends and anteriorly completed with a patch of bovine pericardium and closure of the fistula from the esophageal side with stitches according to the medical report from the center. The patient was doing fine during follow-up, and 1 year later was able to tolerate soft and solid food without dysphagia, as the gastrostomy tube came out by itself a few months after the surgery. Also, he resumed normal physical activities without a neurological sequel. We are unable to do any further endoscopy or contrast studies as the patient has completed follow-up.
Discussion
Ingestion of FBs is common in children younger than 6 years. The most common objects ingested are coins and bones of chicken and fish. Ingestion of BBs accounts for less than 2% of FBs ingested by children [3]. The most harmful and serious complications occur with a battery size of 20 mm [2,4,5]. The injury is attributed to various causes, including electric currents leading to the formation of corrosive hydroxyl ions, and leakage of the alkaline contents of the batteries, causing corrosion and pressure necrosis [4]. While most ingested BBs pass spontaneously without any complications, especially beyond the stomach [3], complications usually occur from BBs that are stuck in the esophagus. The most frequent site of esophageal dislodgment is in the upper esophagus [3,6].
Complications from BB include pneumonia, mediastinitis, hemorrhage, esophageal ulceration, sepsis, tracheoesophageal fistula, perforation of esophagus, tension hydropneumothorax, pneumoperitoneum, and esophageal-vascular fistulas, such as erosion into the subclavian artery or thyroid vessels, AEF, and esophageal stenosis [5,7]. AEF is rare and poses a life-threatening emergency, and it is fatal in most affected patients. Causes of AEF in children include FB ingestion, which mainly include BBs and sharp objects such as pins, and fish and chicken bones. Other FBs, such as vascular rings, coins, and NGT, have been reported, especially in the presence of anatomical abnormalities such as a vascular ring.
Most BB ingestion cases were unwitnessed [2], and the presentation in these patients varied, including non-specific vomiting and vomiting after ingestion of solid and liquid food or only after ingestion of solid food, while liquid intake was tolerated. Additionally, there have also been reports of refusal to eat, dysphagia, odynophagia, sialorrhea, abdominal pain, abdominal distention, hematemesis, melena, fever, irritability, fussy behavior, crying, lethargy, anorexia, respiratory symptoms such as tachypnoea and stridor, dehydration, acrocyanosis, and seizures. A lack of a history of ingestion led to recurrent ER visits and misdiagnosis of upper respiratory tract infection, urinary tract infection, acute gastroenteritis, ear infection, and sepsis [5]. The age of presentation with complications secondary to BB ingestion was reported to be 1-4 years. The duration of development of these complications after ingestion ranged from 10 h to 28 days, with most patients presenting on day 9 or 10 [5].
We report the case of a child who developed AEF 9 days after BB removal and who was resuscitated aggressively. The fistula was diagnosed with CTA. The patient was managed with novel techniques using a vascular duct occluding device that perfectly closed both sides of the fistula and stopped bleeding. This technique works efficiently to save patient until the time of definitive correction. This technique has never previously been reported to treat AEF. In this article, we review the survival and fatal cases regarding demographic characteristics, presentation, mode of diagnosis, and how the surviving cases were managed, including our patient, to show that aggressive rustication and minimally invasive procedure are crucial in saving the patient’s life until definitive correction.
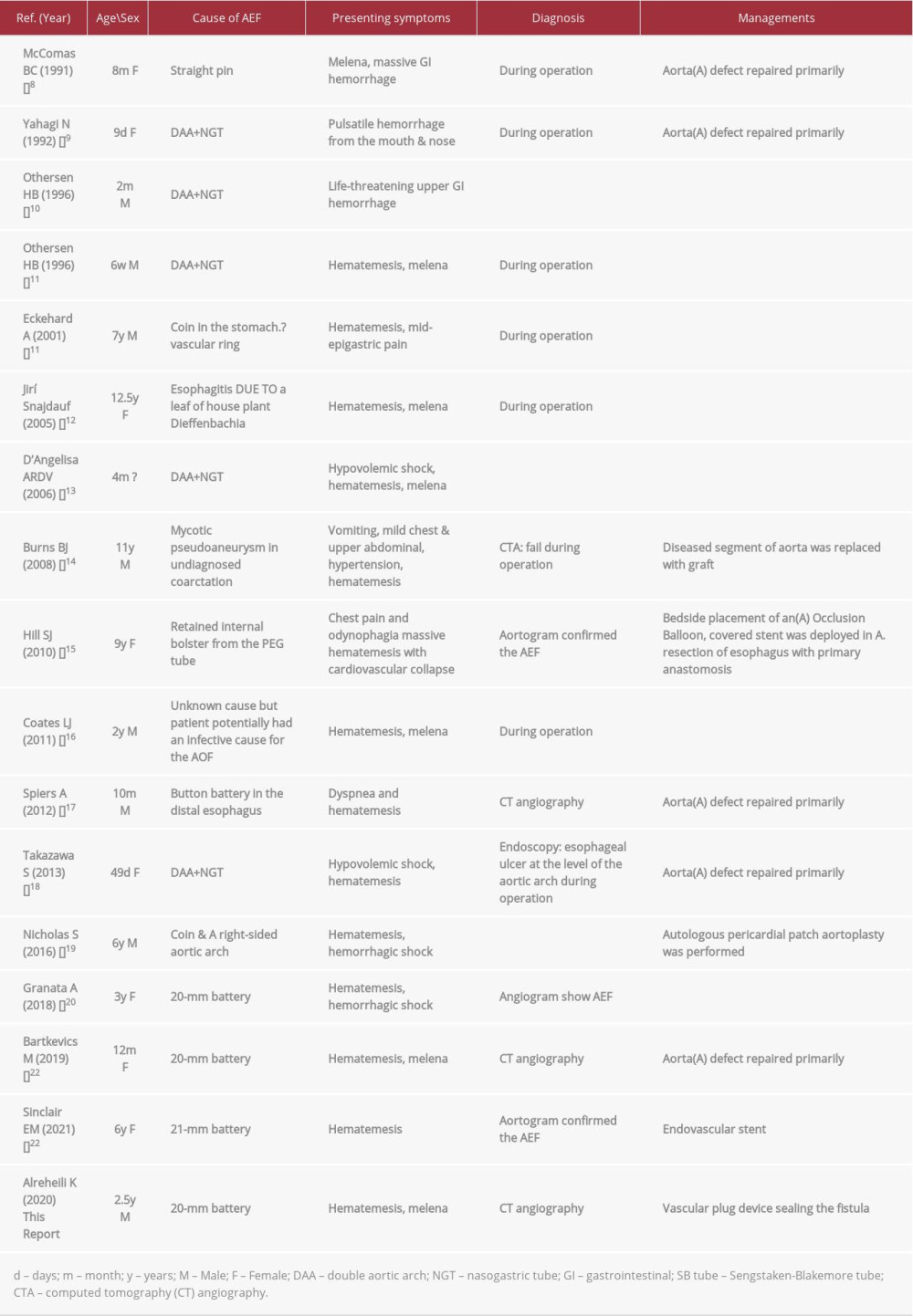
Seventeen patients have been reported to have survived AEF, including our reported patient [8–22]. Table 1 shows a literature review of cases that survived. Five of the cases were secondary to BB, one was due to an unknown cause, potentially an infective cause, one was caused secondary to ingestion of the house plant Dieffenbachia during suicide attempts, and 6 were secondary to a vascular ring with a FB coin, NGT, or ETT. In 3 patients, the cause was a FB (straight pin, a bolster of PEG tube, or a coin), and that in one patient was secondary to mycotic pseudoaneurysm in undiagnosed coarctation of the aorta. In the 5 patients with BB ingestion, the BB was removed 6 h after ingestion in one patient and 14 h in another one while being removed from the stomach, while in the other patient after 36 h of possible ingestion, after 18 h in our patient, and after an unknown duration in the fifth patient, which was removed intraoperatively from the stomach. The BB was discovered in other cases either after laparotomy or after a patient vomited it out, or its ingestion remained unknown. The age of the patients who survived ranged from 9 days to 12.5 years. Presentation was similar to hematemesis and melena or fainting attacks secondary to hypovolemic shock; sentinel exsanguination also ranged from a few hours to a few days. Diagnosis of AEF was performed by computed tomography angiography (CTA) in 3 out of 5 patients and after laparotomy to identify the source of bleeding in 11 patients. Aortography performed in 5 patients failed to detect the fistula in 2 patients. Most patients received aggressive resuscitation with fluid and blood products before successful operation. Regarding management, an esophageal balloon catheter tamponade was performed to control bleeding in 8 out of 17 patients, of whom the Sengstaken-Blakemore tube (SBT) was used in 6, while a Foley balloon catheter was used in one patient, and bedside placement of an aortic occlusion balloon was performed in another patient. Aortic endovascular stent placement was performed in 3 patients. A novel vascular device was done in our patient. In all patients who treated surgically, thoracotomy was performed, the fistula was identified and excised, and esophageal and aortic defects were repaired primarily, except in 2 patients in whom the diseased segment of the aorta was replaced with graft.
A review of published cases was performed using PubMed and the National Capital Poison Canter. Fatal outcomes were reported in 50 cases [5,6,24–42], of which 58% (29 cases) were BB-induced injuries, and 42% (21 cases) were not related to BBs. Among the BB-related cases, 27 were diagnosed with AEF and 2 cases had other vascular erosions. BB size was 20 mm or larger in all cases, except one in which it was 16 mm in size Although our review was up to age 14 years, the age of fatal cases for button battery related cases interestingly ranged from 1 to 4 years. In 22 cases, the AEF was confirmed, and in 5 cases, the diagnosis was made clinically and no diagnostic procedures mentioned in the literature. The BB was removed in 12 cases (11 endoscopic and 1 surgical removal), while removal was unknown in 2 cases. Fatal GI bleeding occurred 10 h to 28 days after removal. Diagnosis of AEF was confirmed postmortem in 15 cases, during the operation in 3 cases, by CTA in one case, and by unknown methods in 3 cases. Non-BB esophago-vascular injuries were reported in 21 cases, all of which were AEF. Ten cases were secondary to vascular rings, 9 of which were double aortic arch (DAA) with prolonged NGT in 9 cases. The duration was reported to be as short as 8 days in pediatric patients, and Montgomery salivary bypass tube (MSBT) was used in one patient. In 6 patients, the AEF was due to FB, and one was due to VATER syndrome with operated TEF and prolonged NGT. One post-PDA repair and iatrogenic sutures in the aorta wall led to subsequent formation of AEF. Two were due to rupture of the mycotic aortic aneurysm and one due to an unknown cause.
Diagnostic procedures included chest and abdominal radiography, esophagography, EGD, CTA, and aortography. The importance of chest and abdominal radiography is during the initial evaluation of the patient. Its importance is to identify FBs as a possible cause of AEF. The stomach may be hugely distended as blood from the fistula accumulates in the stomach and has been reported in many cases. Other findings may indicate rupture of aortic aneurysm or missed coarctation of the aorta, which may be complicated by an aneurysm. These findings include widening of the mediastinum, displacement of the trachea to the right, displacement of the nasogastric tube to the right, and rib notching on chest radiograph. Although barium esophagography has low sensitivity, it may detect AEF in adult patients [43]. We did not find any significant benefit in pediatric patients, which may delay the diagnosis. This is because of the different aetiologias in pediatric and adult patients; the most common cause in adults is thoracic aortic aneurysm, which is rare among children. Endoscopy findings varied in reported cases, and included pulsatile or fresh bleeding [11,12,40], pulsatile submucosal hematoma [44], a non-pulsatile purple mass protruding into the esophagus in the aneurysm [36], esophageal ulcer with [18] or without bleeding [13], thickened mucosa [21], and an odd purple streak [16].
All of these findings were in the middle or upper esophagus. Endoscopy can also be effective in that it can rule out other differential diagnoses. CTA showed extravasation of contrast material from the aorta into the esophagus.
The rate of CTA for AEF in pediatric patients was 60% in our review (Table 1). Rapid diagnosis is critical; therefore, the diagnostic approach for pediatric patients presenting with massive upper gastrointestinal bleeding included upper EGD, CTA, and aortography. The first modality to be performed depended on the most likely diagnosis and the patient’s hemodynamic status. If AEF is suspected, CTA is the preferred initial diagnostic test as it is easier to obtain and less invasive than EGD or aortography. This requires a high index of suspension. Chiari’s triad was used in adult patients [43]. The triad comprises midthoracic pain, sentinel arterial hemorrhage, and exsanguination after a symptom-free interval. We believe it is not applicable to the pediatric age group as almost all patients are under the age of 4 years and they are non-verbalized to indicate pain and to give a history of FB ingestion, as it was most commonly unwitnessed.
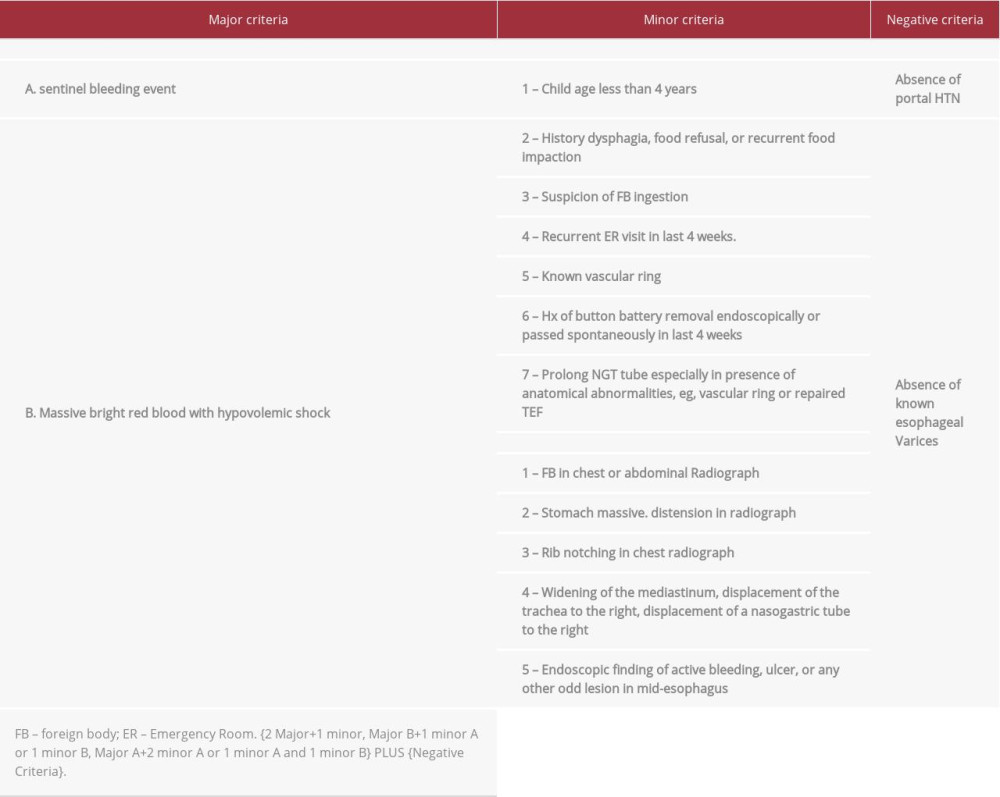
We developed criteria that aided in a high index of suspension for the diagnosis of AEF (Table 2). These criteria were developed depending on the revision of all cases showing survival and fatalities of AEF. It includes 2 major criteria, clinical and radiological/endoscopic minor criteria, and exclusion criteria. Therefore, in the absence of portal hypertension and known cases of esophageal varices, the presence of 2 major+1 minor, major B+1 minor A or 1 minor B, major A+2 minor A or 1 minor A, and 1 minor B will determine a high probability that the patient has AEF. These criteria were applicable for most of reviewed cases and because this is not the scope of this article, we are planning to validate these criteria in the future for all possible reported new cases and do modification as necessary in separate work.
During the management of AEF in children, aggressive resuscitation is crucial to allow diagnostic tests and definitive treatment to be carried out. In many reported cases of survival, patients were supported by large volumes of fluids and blood products. Trials to stop bleeding are also important measures. The SBT has proved advantageous in controlling bleeding. It is valuable for gaining time to allow for definitive surgical treatment. Percutaneous endovascular aortic repair (EVAR) using balloon expandable stents is used in pediatric patients. Three patients have been reported to survive [15,20], while in one patient with mycotic aneurysms, the definitive surgical treatment was delayed, leading to death of the patient 3 months after stent placement [35]. In adult studies, EVAR should be considered only as a temporizing strategy until definitive repair can be performed [45]. In addition, the use of EVAR in children is debatable because of technical considerations, and it is recommended as a lifesaving procedure. In our case, we report the novel use of a vascular plug device to close both aortic and esophageal defects and which was stable to date, almost 2 month later. Whether to consider this procedure as a definitive treatment or as a bridge to definitive surgical repair needs to be evaluated. Surgical procedures must consider the procedure of choice at the time, and must include identification and resection of the fistula and repair of esophageal and aortic defects with or without the use of a graft.
Conclusions
AEF is rare and has a fatal outcome in most cases. Diagnosis requires a high index of suspicion, and treatment should include aggressive management. The endovascular device remains a non-invasive emergency treatment option and should be considered as lifesaving and as a bridge to definitive repair.
Figures
References:
1.. Mubarak A, Benninga MA, Broekaert I, Diagnosis, management, and prevention of button battery ingestion in childhood: A European Society for Paediatric Gastroenterology Hepatology and Nutrition position paper: J Pediatr Gastroenterol Nutr, 2021; 73(1); 129-36
2.. Litovitz T, Whitaker N, Clark L, Preventing battery ingestions: An analysis of 8648 cases: Pediatrics, 2010; 125(6); 1178-83
3.. Yardeni D, Yardeni H, Coran AG, Golladay ES, Severe esophageal damage due to button battery ingestion: Can it be prevented?: Pediatr Surg Int, 2004; 20(7); 496-501
4.. Litovitz T, Schmitz BF, Ingestion of cylindrical and button batteries: An analysis of 2382 cases: Pediatrics, 1992; 89(4 Pt 2); 747-57
5.. , National Capital Poison Center Fatal Button Battery Ingestions: 62 Reported Cases . Accessed August 8, 2020http://www.poison.org/battery/fatalcases
6.. Anand S, Jain V, Agarwala S, Esophageal button battery in the pediatric population: Experience from a tertiary care center: Indian J Pediatr Aug, 2020; 87(8); 591-597
7.. Leinwand K, Brumbaugh DE, Kramer RE, Button battery ingestion in children: A paradigm for management of severe pediatric foreign body ingestions: Gastrointest Endosc Clin N Am, 2016; 26(1); 99-118
8.. McComas BC, van Miles P, Katz BE, Successful salvage of an 8-month-old child with an aortoesophageal fistula: J Pediatr Surg, 1991; 26(12); 1394-95
9.. Yahagi N, Nishikawa A, Sai Y, Double aortic arch presenting as massive haematemesis after removal of a nasogastric tube: Can J Anaesth, 1992; 39(8); 894
10.. Othersen HB, Khalil B, Zellner J, Aortoesophageal fistula and double aortic arch: Two important points in management: J Pediatr Surg, 1996; 31(4); 594-95
11.. Stuth EA, Stucke AG, Cohen RD, Successful resuscitation of a child after exsanguination due to aortoesophageal fistula from undiagnosed foreign body: Anesthesiology, 2001; 95(4); 1025-26
12.. Snajdauf J, Mixa V, Rygl M, Aortoesophageal fistula – an unusual complication of esophagitis caused by Dieffenbachia ingestion: J Pediatr Surg, 2005; 40(6); e29-31
13.. D’Angelis AR, Questa H, Prieto F, Successful surgical treatment of a 4-month infant after exsanguination for aortoesophageal fistula: J Pediatr Surg, 2006; 41(4); 848-49
14.. Burns BJ, Newey A, Numa A, Beware the starboard nasogastric tube: Pediatr Emerg Care, 2008; 24(5); 307-9
15.. Hill SJ, Zarroug AE, Ricketts RR, Veeraswamy R, Bedside placement of an aortic occlusion balloon to control a ruptured aorto-esophageal fistula in a small child: Ann Vasc Surg, 2010; 24(6); 822.e7-9
16.. Coates LJ, McNally J, Caputo M, Cusick E, Survival in a 2-year-old boy with hemorrhage secondary to an aortoesophageal fistula: J Pediatr Surg, 2011; 46(12); 2394-96
17.. Spiers A, Jamil S, Whan E, Survival of patient after aorto-oesophageal fistula following button battery ingestion: ANZ J Surg, 2012; 82(3); 186-87
18.. Takazawa S, Uchida H, Kawashima H, Massive hemorrhage after Kasai portoenterostomy in a patient with a congenital extrahepatic portosystemic shunt, malrotation and a double aortic arch: Report of a case: Surg Today, 2014; 44(8); 1561-64
19.. Clarke NS, Murthy R, Hernandez J, Aortoesophageal fistula in a child with undiagnosed vascular ring: Life-threatening or lethal?: Ann Thorac Surg, 2016; 102(4); e325-27
20.. Granata A, Gandolfo C, Acierno C, Button battery removed from the stomach resulting in a missed aortoesophageal fistula – a multidisciplinary approach to rescuing a very young patient: A case report: J Med Case Rep, 2018; 12(1); 318
21.. Bartkevics M, Stankovic Z, Schibli S, A near miss and salvage management of aortoesophageal fistula secondary to cell battery ingestion: World J Pediatr Congenit Heart Surg, 2020; 11(1); 120-22
22.. Sinclair EM, Stevens JP, McElhanon B, Development and repair of aorto-esophageal fistula following esophageal button battery impaction: A case report: J Pediatr Surg Case Rep, 2021; 66; 101782
23.. Karnecki K, Pieśniak D, Jankowski Z, Fatal haemorrhage from an aortoesophageal fistula secondary to button battery ingestion in a 15-month-old child. Case report and literature review: Leg Med (Tokyo), 2020; 45; 101707
24.. Loots DP, du Toit-Prinsloo L, Saayman G, Disk battery ingestion: A rare cause of perforation of the brachiocephalic artery: Forensic Sci Med Pathol, 2015; 11(4); 614-17
25.. Brumbaugh DE, Colson SB, Sandoval JA, Management of button battery-induced hemorrhage in children: J Pediatr Gastroenterol Nutr, 2011; 52(5); 585-89
26.. Mortensen A, Hansen NF, Schiødt OM, Fatal aortoesophageal fistula caused by button battery ingestion in a 1-year-old child: Am J Emerg Med, 2010; 28(8); 984.e5-6
27.. Hoshina T, Nomura K, Ko Y, [hemorrhagic shock due to an aortoesophageal fistula in an infant with a double aortic arch; report of a case]: Kyobu Geka, 2015; 68(13); 1085-88 [in Japanese]
28.. Atsumi N, Matsubara M, Kimura N, Terada M, Fatal aortotracheal fistula combined with aortoesophageal fistula in an infant with double aortic arch: A warning: Gen Thorac Cardiovasc Surg Oct, 2015; 63(10); 579-82
29.. Sigalet DL, Laberge JM, DiLorenzo M, Aortoesophageal fistula: Congenital and acquired causes: J Pediatr Surg, 1994; 29(9); 1212-14
30.. McKeating J, Smith S, Kochanck P, Fatal aortoesophageal fistula due to double aortic arch: An unusual complication of prolonged nasogastric intubation: J Pediatr Surg, 1990; 25(12); 1298-300
31.. Grey TC, Mittleman RE, Wetli CV, Horowitz S, Aortoesophageal fistula and sudden death. A report of two cases and review of the literature: Am J Forensic Med Pathol, 1988; 9(1); 19-22
32.. Pehlivan S, Kara DO, Turkkan D, Fatal aorto-esophageal fistula in child: a case report: J Forensic Leg Med, 2014; 22; 112-14
33.. McWhorter V, Dunn JC, Teitell MA, Aortoesophageal fistula as a complication of Montgomery salivary bypass tube: J Pediatr Surg, 2005; 40(4); 742-44
34.. van Woerkum F, Bont L, Haas F, Aortoesophageal fistula due to double aortic arch and prolonged nasogastric intubation: Case report and review of the literature: Eur J Pediatr, 2006; 165(9); 660-61
35.. Alsaadi A, Arabi M, Bashir O, Percutaneous endovascular aortic repair with left carotid chimney for arch mycotic aneurysm and aortoesophageal fistula in a pediatric patient: J Vasc Interv Radiol, 2019; 30(10); 1693-95
36.. Sheiko MA, Hoffenberg EJ, Massive hematemesis as presentation of congenital aortic coarctation with superinfected aneurysm and aortoesophageal fistula: J Pediatr Gastroenterol Nutr, 2016; 62(1); e4-5
37.. Maarouf Z, Kajo K, Plank L, Secondary aortoesophageal fistula as a lethal complication of continuous nasogastric sondage in a child with VATER syndrome: Ann Saudi Med, 2001; 21(3–4); 213-15
38.. Angelini A, Dimopoulos K, Frescura C, Fatal aortoesophageal fistula in two cases of tight vascular ring: Cardiol Young, 2002; 12(2); 172-76
39.. Heck HA, Moore HV, Lutin WA, Esophageal-aortic erosion associated with double aortic arch and tracheomalacia. Experience with 2 infants: Tex Heart Inst J, 1993; 20(2); 126-29
40.. Al Lawati TT, Al Brashdi Y, Al Rahbi N, Sengstaken-Blakemore tube: An extra mile: Int J Pediatr Adolesc Med, 2017; 4(4); 141-43
41.. Takesaki NA, Reis MC, Miranda ML, Baracat EC, Hemorrhagic shock secondary to button battery ingestion: Sao Paulo Med J, 2014; 132(3); 184-88
42.. el Barbary AS, Foad H, Fathi A, Oesophageal fistulae caused by swallowed foreign bodies: J Laryngol Otol, 1969; 83(3); 251-59
43.. Hollander JE, Quick G, Aortoesophageal fistula: A comprehensive review of the literature: Am J Med, 1991; 91(3); 279-87
44.. Naschitz JE, Bassan H, Lazarov N, Grishkan A, Upper gastrointestinal bleeding, radiographic, and endoscopic features: J Clin Gastroenterol, 1987; 9; 342-44
45.. Antoniou GA, Koutsias S, Antoniou SA, Outcome after endovascular stent graft repair of aortoenteric fistula: A systematic review: J Vasc Surg, 2009; 49(3); 782-89
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133