22 July 2021: Articles 
Unusual Movement Disorders and Atypical Magnetic Resonance Imaging (MRI) Findings in Patients with West Nile Encephalitis: Case Reports of 2 Patients with Evidence of Clinical and Imaging Resolution with IVIG
Unusual clinical course, Challenging differential diagnosis, Unusual or unexpected effect of treatment, Patient complains / malpractice, Rare disease, Educational Purpose (only if useful for a systematic review or synthesis), Rare coexistence of disease or pathology
Irfan S. Sheikh1ABCDEF*, Ehad Afreen1AF, Ajaz Sheikh1ADEDOI: 10.12659/AJCR.932215
Am J Case Rep 2021; 22:e932215
Abstract
BACKGROUND: West Nile virus (WNv) is the leading cause of epidemic arbovirus encephalitis in the continental United States. Movement disorders (MDs) have been reported in 20% to 40% of patients with WNv and about 37% of patients with WNv encephalitis have changes on magnetic resonance imaging (MRI). We report 2 unusual cases of neuroinvasive WNv in patients with unusual MDs and unreported MRI findings.
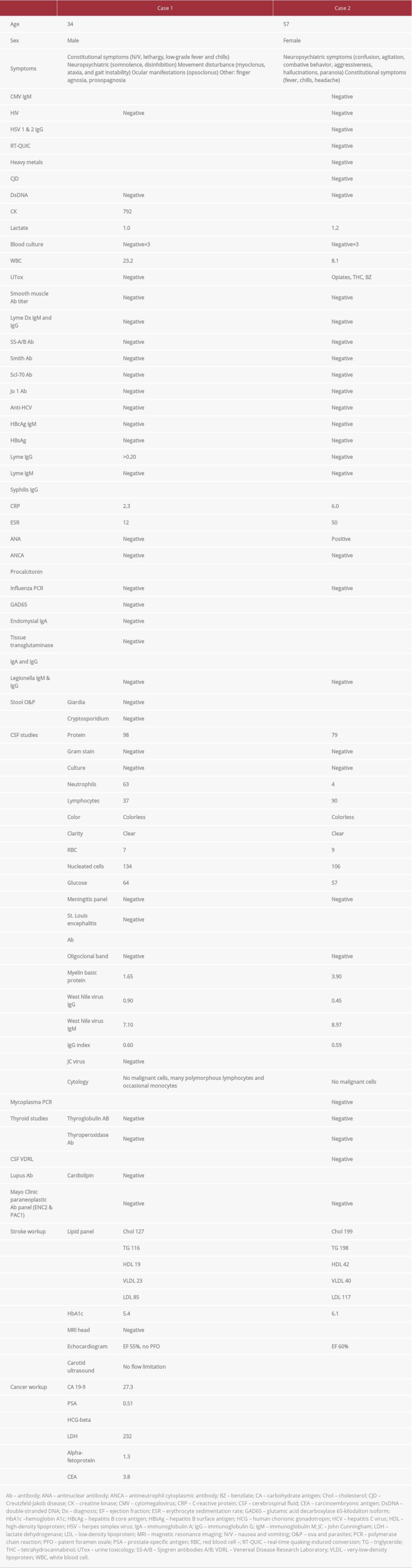
CASE REPORT: In the first case, a 34-year-old man presented with a 1-week history of disinhibition, agitation, opsoclonus-myoclonus and ataxia syndrome (OMAS), tremor, and facial agnosia. Evaluation of his cerebrospinal fluid (CSF) revealed elevated immunoglobulin (Ig)M against WNv, a high level of protein (98 mg/dL), and an elevated white blood cell (WBC) count (134, 37% lymphocytes). An MRI of the brain showed an area of diffusion restriction in the splenium of the corpus callosum. The patient’s MRI findings and OMA improved significantly after 2 treatments with i.v. IG (IVIG). In the second case, a 57-year-old woman presented with fever, headaches, psychosis, and ataxia; she was subsequently intubated for airway protection. Analysis of her CSF showed elevated IgM against WNv, a high level of protein (79 mg/dL), and elevated WBC count (106, 90% lymphocytes). One week after the onset of symptoms, the patient experienced facial dyskinesia. Later, she developed proximal bilateral lower extremity weakness. An MRI of her lumbar spine showed evidence of myeloradiculitis with contrast enhancement of the conus medullaris and ventral nerve roots. After a single treatment with IVIG, she had partial improvement in weakness.
CONCLUSIONS: MDs and changes on MRI have been reported in patients with neuroinvasive WNv disease. Our patient with OMAS also had transient splenial diffusion restriction on imaging, which, to the best of our knowledge, has not been previously reported with WNv infection. In both patients, treatment with IVIG resulted in improvement in symptoms.
Keywords: Corpus Callosum, Dyskinesias, Opsoclonus-Myoclonus Syndrome, radiculopathy, West Nile virus, Immunoglobulins, Intravenous, Magnetic Resonance Imaging, Movement Disorders, United States, West Nile Fever
Background
West Nile virus (WNv) is the leading cause of epidemic arbo-virus encephalitis in the continental United States [1]. It is a zoonotic arbovirus that belongs to the
Case Reports
CASE 1:
A 34-year-old right-handed man presented to the University Hospital with a 1-week history of nausea, vomiting, lethargy, low-grade fever and chills, somnolence, disinhibition, jerking movements of his upper extremities, an inability to recognize familiar people or hands, and gait instability. His physical exam was significant for facial and finger agnosia; rapid, involuntary, multivectoral (horizontal and vertical), and conjugate fast eye movements, with intersaccadic intervals associated with myoclonus (opsoclonus); left knee flexion weakness (Medical Research Council grades 4–5); extensor plantar response in the left foot; right finger flexion response (positive Hoffman sign); bilateral finger-to-nose dysmetria; a positive Brudzinski sign; and gross appearance of periodic, fast, myoclonic jerking movements of the left upper and right lower extremities in an asynchronous arrhythmical pattern.
The patient was admitted to the Intensive Care Unit (ICU) for careful monitoring and started on prophylactic antibiotics and antiviral treatment for suspected meningoencephalitis because initial testing of cerebrospinal fluid (CSF) showed signs of inflammation (Table 1). Serum laboratory testing revealed elevation in creatine kinase (792 U/L) and leukocytosis (23.2×109/L). An MRI of the brain with and without contrast revealed a 0.4-mm focus of diffusion restriction in the corpus callosum with reversal on apparent diffusion coefficient, suggestive of cytotoxic edema (Figure 1). A workup for stroke was unrevealing. Given the patient’s clinical presentation and the objective evidence, he was diagnosed with post-infectious OMAS.
Over the next several days, the patient’s clinical status deteriorated rapidly. After a discussion about the risks and benefits of treatment with IVIG, he was started on 1 g/kg i.v. for 2 days. It was repeated after 4 days because the first treatment resulted in limited clinical improvement. After receiving IVIG, the patient had moderate improvement in symptoms.
At this point, because the etiology of the patient’s symptoms was still unknown, further workup was ordered and the results were unremarkable (Table 1). On day 7 of the patient’s hospital stay, his CSF tested positive for West Nile IgM antibodies, with titers at 7.10. On day 11, a repeat MRI of the brain with contrast enhancement showed interval resolution of the focal restricted diffusion in the corpus callosum (Figure 2). The patient was discharged to inpatient rehab after a 13-day hospital stay. When he was seen in the clinic 1 year later, his symptoms had resolved completely and a neurological examination revealed no focal findings.
CASE 2:
A 57-year-old woman presented with a 3-day history of confusion, combativeness, low-grade fever, headaches, tremors, and nausea and vomiting with acute onset. Her symptoms had begun with mild tremors that occurred at rest and with intention, followed by fevers, chills, combativeness, and aggression with violent outbursts against others and inanimate objects. This progressed to acute psychosis with visual hallucinations (seeing people who were not there), along with paranoia, with progression of the headaches. She was being treated for bipolar disorder with lithium and lamotrigine and was using marijuana recreationally.
Physical examination of the patient was significant for agitation, combativeness, disorientation, dysarthria, and perseverative speech. She also exhibited a right gaze preference, sialorrhea, and nuchal rigidity (positive Brudzinski sign). She was transferred to the ICU and intubated for airway protection. An initial MRI of the brain was unremarkable. Her serum and CSF were tested (Table 1). The CSF was positive for high titers of WNv IgM I (8.97).
One week after her presentation and following extubation, the patient developed facial dyskinesia, including continuous perioral puckering, lip smacking, and mouth opening. An electroencephalogram was done to capture these events, but it was negative for any epileptiform activity and showed diffuse slowing. Cognitively, the patient showed improvement, although she was still mildly encephalopathic. However, she had signs of lower-extremity weakness. An MRI of her thoracic and lumbar spine (Figure 3) with and without contrast showed leptomeningeal enhancement along the conus medullaris and multiple ventral nerve roots. There were also patchy areas of edema and enhancement at the T10 to T12 endplates, with involvement of the anterior horn cell/root in the thoracolumbar region. This was thought to be associated with the WNv infection. A repeat lumbar puncture showed a reduction in nucleated cells down to 31 and a slight increase in the percentage of CSF lymphocytes to 94%. The patient’s myeloradiculitis was determined to be secondary to WNv infection and she was started on IVIG for 5 days, to a total of 2 g/kg. After a single treatment with IVIG, there was partial improvement in her weakness. Over the remaining course of the patient’s hospital stay, her weakness continued to improve but her mental status fluctuated. She had episodes of psychosis, including paranoia and delusions. She exhibited symptoms of mania and was evaluated by a psychiatrist. Her outpatient prescription for lithium was adjusted and she was started on risperidol. She did not respond adequately and was eventually transferred for further inpatient psychiatric treatment.
Discussion
Most patients with WNv infection never have symptoms; 80% of cases are asymptomatic [2]. The most common symptoms are influenza-like, including fever, malaise, headache, joint pain, and lymphadenopathy, along with gastrointestinal manifestations such as nausea, vomiting, and decreased appetite. Other manifestations can include skin eruption (punctate exanthema, macula or papules), hepatomegaly, splenomegaly, and, occasionally, myocarditis and pancreatitis [12]. Central nervous system (CNS) involvement is uncommon and, when present, can consist of encephalitis, meningitis, Guillain-Barré Syndrome, optic neuritis, poliomyelitis-like paralysis, and, rarely, MDs, including OMAS [13]. Other MDs have been associated with WNv, but in the presence of encephalitis [8]. Our patient in Case 2 presented with WNv encephalitis but subsequently developed abnormal facial dyskinesia, which to the best of our knowledge is the first such report in the literature.
Diagnosis of WNv is multimodal and often requires analysis of findings from both imaging and serum. MRI findings in neuroinvasive WNv have been shown to involve diffusion-weighted changes in subcortical white matter, corona radiata, and the internal capsule; T2-hyperintense flairs in gray and white matter, the cerebellum, basal ganglia, thalamus and internal capsule, pons and midbrain; and enhancement of the meninges, spinal cord, cauda equina, and nerve roots [11]. Infections are a known cause of corpus callosum diffusion restriction on MRI. Such infections include EBV, herpes simplex virus, VZV,
The treatment of WNv encephalitis and neuroinvasive disease is mainly supportive. In mouse models, studies have shown a benefit for IVIG; pooled human WNV convalescent sera (WNV-IVIG) also inhibited development of lethal WNV encephalitis by suppressing CNS infiltration by CD45(high) leukocytes [15]. That study also showed that IVIG profoundly reduced the infiltration of Ly6C(high), CD11b-positive monocytes, which were the major infiltrating subset in the CNS of infected control mice [6]. The findings confirm the potent immunomodulatory activity of generic IVIG and underscore its potential as an effective immunotherapeutic drug for encephalitis and other virus-induced inflammatory diseases [15].
Subsequent clinical trials have shown limited efficacy for IVIG in treatment of WNv encephalitis [16]. In the first trial, 242 patients were screened, but only 64 (26%) met the entry criteria; therefore, their data were not tabulated. This trial was terminated prematurely because of slow accrual and reduced availability of study products. The trial also did not look at use of IVIG in patients with WNv-induced OMAS or atypical MDs. A study published in Israel in 2012 [17] showed a prompt response to treatment with IVIG in some patients with neuroinvasive WNv disease. The findings were consistent with the responses observed in 10 patients that were previously reported; the Israeli genotypes were highly homologous to American viruses and with cases reported outside of Israel. In this study, the population of patients given IVIG also was limited; however, 3 of them made a complete recovery.
Given the data and studies available, we administered IVIG after a full discussion of the risks and benefits with our patients, who were deteriorating rapidly. Both of them were treated with IVIG. In Case 1, the result was a more rapid resolution of objective findings, with resolution on MRI imaging within 1 week after initiation of therapy. It is important to note that treatment began on hospital day 2 and was started empirically, based on the patient’s clinical presentation and objective data, including preliminary results of studies of CSF and MRI findings, but before serology results confirming WNv were available. If IVIG is considered in a patient who is rapidly deteriorating and has WNv-induced OMAS or an atypical MD, the risks and benefits should be thoroughly discussed with the patient before administration. There are also studies showing that monoclonal antibodies are effective in this clinical setting [18,19].
Various MDs have been recognized with neuroinvasive WNv infection. Frequently reported MDs include tremors, extrapyramidal symptoms, myoclonus, choreoathetosis, and hemiballismus. The facial dyskinesia seen in the patient in Case 2 has yet to be extensively reported in the literature. Our patient had a history of bipolar disorder, but a thorough review of records from her pharmacy and of the medication administered to her in the hospital showed that she was not on any antipsychotic, antiemetic, or known anti-dopaminergic medication that could have precipitated the dyskinesia. Her only home medication for bipolar disorder was lithium, which was confirmed by her outpatient psychiatrist. Furthermore, her dyskinesia was transient and had all but completely resolved by the time of discharge. While the patient was in the hospital, she developed psychosis after the dyskinesia had resolved, which was secondary to under-dosing of her lithium, for which a psychiatrist was consulted. She was put on risperidone 1 day before discharge and did not have any extra-pyramidal adverse effects as a result.
The mechanism of the cytotoxic lesion in the corpus callosum and OMAS in neurodegenerative WNv is not entirely clear. However, literature on the suspected pathogenesis of opsoclonus alludes to the possibility of damage to the omnipause cells in the nucleus raphe interpositus of the pons [20] or dis-inhibition of the oculomotor region of the fastigial nucleus by a process that interferes with the inhibitory projections of the Purkinje cells of the deep cerebellar nuclei to the fastigial nuclei [20]. When an autoimmune etiology is suspected, the literature supports autoimmune dysfunction of the Purkinje cells in the dorsal vermis and subsequent disinhibition of the oculomotor fastigial region [21]. Mechanisms underlying opsoclonus that occurs after infection are probably related to dys-function in cell- and humoral-mediated immune responses [4]. This has been reported to occur after infection with entero-virus,
The prognosis for patients who have neuroinvasive WNv is variable and often depends on other comorbid and immuno-deficiency states; the mortality rate is estimated to be 10% to 30% [23]. Neurological recovery is estimated to take 6 months to 1 year and can be incomplete, with persistence of headache, muscle pain, lethargy, and asthenia [24]. The patient in Case 2 had baseline neuropsychiatric illness (bipolar disorder), which could explain her incomplete recovery and residual psychiatric symptoms even after resolution of her other symptoms. The patient with OMAS in Case 1, who had limited comorbidities, responded very quickly to IVIG, with improvement in clinical and radiologic signs.
Because neuroinvasive disease is uncommon with WNv, clinical trials of IVIG or plasmapheresis for it have not been performed. Thus, more studies are needed to validate use of IVIG for neuroinvasive WNv, along with studies to show a correlation between IVIG and resolution of changes on MRI.
Conclusions
Neuroinvasive WNv can cause significant morbidity in affected patients. Unusual MRI findings have been reported in patients with it. This disease process also should be considered when a cytotoxic lesion of the corpus callosum is present. MDs also have been seen in patients with neuroinvasive WNv, and when these individuals are encountered, careful attention to facial dyskinesia can lead to suspicion of an underlying arbo-virus illness, such as WNv. Our case reports advance knowledge about neuroinvasive WNv, expand upon what is already known in the literature, and provide new insight into uncommon and unusual presentations of this disease.
Figures
References:
1.. Curren EJ, Lehman J, Kolsin J, West Nile virus and other nationally notifiable arboviral diseases-United States, 2017: MMWR Morb Mortal Wkly Rep, 2018; 67; 1137-42
2.. Birlutiu V, Birlutiu RM, Rezi EC, Infection with West Nile Virus: Opsoclonusmyoclonus syndrome: Research on Immunology, 2016 978-1-922227-91-1
3.. Klaas JP, Ahlskog JE, Pittock SJ, Adult-onset opsoclonus-myoclonus syndrome: Arch Neurol, 2012; 69(12); 1598-607
4.. Ertekin V, Tan H, Opsoclonus-myoclonus syndrome attributable to hepatitis C infection: Pediatr Neurol, 2010; 42(6); 441
5.. Medrano V, Royo-Villanova C, Flores-Ruiz JJ, [Parainfectious opsoclonus-myoclonus syndrome secondary to varicella-zoster virus infection.]: Rev Neurol, 2005; 41(8); 507-8 [in Spanish]
6.. Borg M, Symptomatic myoclonus: Neurophysiol Clin, 2006; 36(5–6); 309-18
7.. Deconinck N, Scaillon M, Segers V, Opsoclonus-myoclonus associated with celiac disease: Pediatr Neurol, 2006; 34(4); 312-14
8.. Gyure KA, West Nile virus infections: J Neuropathol Exp Neurol, 2009; 68(10); 1053-60
9.. Birlutiu V, Birlutiu RM, Opsoclonus-myoclonus syndrome attributable to West Nile encephalitis: A case report: J Med Case Rep, 2014; 8; 232
10.. Bataller L, Graus F, Saiz A, Vilchez JJ, Clinical outcome in adult onset idiopathic or paraneoplastic opsoclonus-myoclonus: Brain, 2001; 124; 437-43
11.. Ali M, Safriel Y, Sohi J, Llave A, Weathers S, West Nile Virus infection: MR imaging findings in the nervous system: Am J Neuroradiol, 2005; 26; 289-97
12.. Goldblum N, West Nile Fever: The clinical features of the disease and the isolation of West Nile virus from the blood of nine human cases: Am J Hyg, 1954; 59; 89-103
13.. Burton JM, Neurological manifestations of West Nile virus infection: Can J Neurol Sci, 2004; 31(2); 189-93
14.. Starkey F, Kobayashi N, Numaguchi Y, Moritani T, Cytotoxic lesions of the corpus callosum that show restricted diffusion: Mechanisms, causes and manifestations: Radiographics, 2017; 37(2); 562-76
15.. Srivastava R, Ramakrishna C, Cantin E, Anti-inflammatory activity of intravenous immunoglobulins protects against West Nile virus encephalitis: J Gen Virol, 2015; 96; 1347-57
16.. Gnann JW, Agrawal A, Hart J, Lack of efficacy of high-titered immunoglobulin in patients with West Nile virus central nervous system disease: Emerg Infect Dis, 2019; 25(11); 2064-73
17.. Shimoni Z, Bin H, Bulvik S, The clinical response of West Nile virus neuroinvasive disease to intravenous immunoglobulin therapy: Clin Pract, 2012; 2(1); e18
18.. Oliphant T, Development of humanized monoclonal antibody with therapeutic potential against West Nile virus: Nat Med, 2005; 11; 522-30
19.. Morrey JD, Defining limits of humanized neutralizing monoclonal antibody treatment for West Nile virus neurological infection in a hamster model: Antimicrob Agents Chemother, 2007; 51; 2396-402
20.. Wong A, An update on opsoclonus: Curr Opin Neurol, 2007; 20; 25-31
21.. Morales-Briceño H, Rodríguez-Violante M, Cervantes-Arriaga A, Opsoclonus myoclonus syndrome associated with GQ1b antibodies: Mov Disord, 2012; 27(13); 1615-16
22.. Yiu VW, Kovithavongs T, McGoningle LF, Ferrerira P, Plasmapheresis as an effective treatment for opsoclonus-myoclonus syndrome: Pediatr Neurol, 2001; 24; 72-74
23.. Burton JM, Neurological manifestations of West Nile virus infection: Can J Neurol Sci, 2004; 31(2); 189-93
24.. Sejvar JJ, Neurologic manifestations and outcome of West Nile virus infection: JAMA, 2003; 290(4); 511-15
Figures
In Press
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942770
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943214
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943010
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943687
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250