07 August 2021: Articles 
Pleural Effusion Secondary to Dasatinib Following Allogenic Hematopoietic Stem Cell Transplantation
Unusual clinical course, Challenging differential diagnosis, Diagnostic / therapeutic accidents, Unusual setting of medical care, Rare disease, Adverse events of drug therapy, Educational Purpose (only if useful for a systematic review or synthesis)
Riyadh R. Alrubaye1ABCDEF*, Celine A. Fadel2DEF, Comfort Y. Adewunmi2ABCD, Loida Del Rio Lopez2ABFDOI: 10.12659/AJCR.932711
Am J Case Rep 2021; 22:e932711
Abstract
BACKGROUND: Pleural effusions are frequently seen among patients with hematopoietic stem cell transplantation (HSCT). In the majority of cases, they are related to infections and volume overload. Medications have also been reported to cause pleural effusion in the general population, albeit very rarely. Dasatinib-induced pleural effusion has been reported in patients with chronic myeloid leukemia but not in those with HSCT. We here report a case of dasatinib-induced pleural effusion following HSCT for acute lymphocytic leukemia (ALL). The proposed mechanism of dasatinib-induced pleural effusion involves build-up of fluid due to an immune-mediated vascular insult.
CASE REPORT: A 72-year-old man who received HSCT for ALL was treated with dasatinib to prevent a recurrence. After 6 months, the patient was admitted to the hospital for pneumonia, which was observed as bilateral pleural effusion upon chest X-ray. After completing the antibiotics course, he developed recurrent pleural effusion during hospitalization. Repeated thoracentesis of the fluid revealed an exudative lymphocytic effusion with negative culture and cytology. Dasatinib was withdrawn and the pleural effusion resolved gradually.
CONCLUSIONS: In patients with dasatinib-induced pleural effusions following HCTS, withdrawal of the drug leads to symptom resolution, thereby avoiding unnecessary procedures. This case illustrates that dasatinib-induced pleural disease typically manifests with lymphocytic exudative fluid. Physicians should be aware that pleural effusion is a possible medication-related adverse effect, which may be missed in cases of infection in patients following HSCT.
Keywords: Bone Marrow Transplantation, dasatinib, Drug-Related Side Effects and Adverse Reactions, Medication Reconciliation, Pleural Effusion, Pyrimidines, Thiazoles
Background
Pleural effusions are common in patients who are undergoing hematopoietic stem cell transplantation (HSCT), with a reported incidence of approximately 10% based on a large retrospective study [1]. The common causes of pleural effusion are infection and volume overload. Rare causes of pleural effusion include malignancy and graft versus host disease (GVHD). Medication-induced pleural effusion among patients undergoing HSCT has not been reported previously. Nevertheless, there is a higher incidence of pleural effusion in patients that are taking medications such as tacrolimus and mycophenolate as prophylaxis against GVHD [1].
The incidence of dasatinib-induced pleural effusion in chronic myeloid leukemia (CML) ranges from 15–30% [2]. Dasatinib was first approved to treat CML but can also be used to prevent acute lymphocytic leukemia (ALL) relapse following HSCT [3]. Here, we report a case of dasatinib-induced recurrent pleural effusion in a patient with ALL following HSCT. To the best of our knowledge, this is the first case of this adverse effect in a patient who has undergone HSCT.
Case Report
A 72-year-old man was diagnosed with Philadelphia chromo-some-positive B-type ALL, through the detection of the peripheral
After treatment with dasatinib and during HSCT, the patient’s cerebral spinal fluid findings were normal, and no
Six months after HSCT, the patient presented with a 10-day history of worsening shortness of breath and a cough. He did not report any orthopnea, paroxysmal nocturnal dyspnea, leg edema, or weight change. He also had a fever that had started the previous day. No complications following HSCT were noted during periodic follow-up visits to an oncologist. He was a nonsmoker with no history of congestive heart failure or tuberculosis. His home medications included dasatinib (100 mg orally daily), sirolimus (1 mg orally daily), and acyclovir (one 400-mg oral tablet daily). His vaccinations were up to date, and he had recently received a
On physical clinical examination, the patient was febrile with a temperature of 39.5°C. He had tachycardia, tachypnea, and mild hypoxia with oxygen saturation of 89% on room air; there was no leg edema. He had bilateral course crackles with poor air entry bilaterally. His abdomen was soft on palpation and no ascites were noted.
Blood tests indicated a hemoglobin level of 10.5 gm/dL, mild acute kidney injury with a creatinine level of 1.5 mg/dL, and mild elevation of his liver enzyme levels. A chest radiograph and chest computed tomography (Figures 1, 2) showed a bilateral pleural effusion with possible left-sided infiltration or pneumonia. A transthoracic echocardiogram showed normal left ventricular function with a small pericardial effusion. Pneumonia, bilateral pleural effusion, and small pericardial effusion were diagnosed in the patient. He was started on treatment with broad-spectrum antibiotics, including vancomycin (1.5 mg every 12 h), cefepime (1 mg intravenously every 8 h), and metronidazole (500 mg intravenously every 8 h) for coverage of anerobic bacteria.
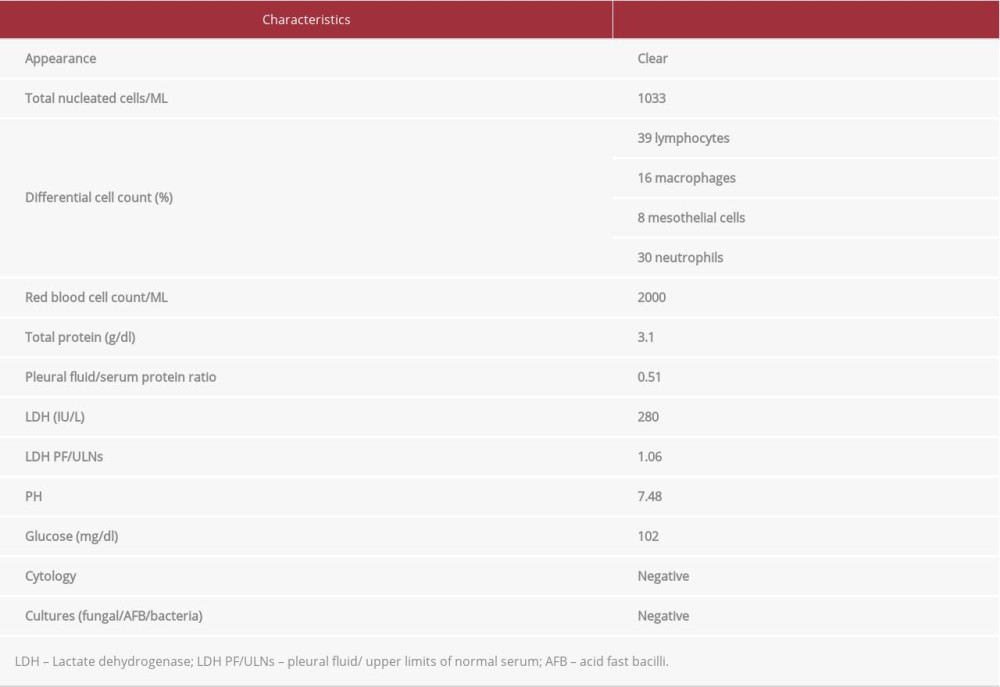
The patient’s fever resolved on the second day of hospitalization, and his symptoms improved. His creatinine and liver enzyme levels also improved. However, he had significant hypoxia with an increased oxygen requirement. Bilateral thoracentesis was performed to drain the pleural fluid, with approximately 1.5 L of fluid drained from each side. The results of the analysis of the pleural fluid are summarized in Table 1. The patient received a diagnosis of left-sided pneumonia and a bilateral exudative pleural effusion according to Light’s criteria (Figure 3) with lymphocytic predominance. The pleural fluid culture and cytology were negative. All investigations for infection, including a blood culture, serum
Despite completing a 5-day course of antibiotics, the patient continued to require oxygen supplementation; therefore, we decided to repeat the chest computed tomography, which showed reaccumulation of the pleural fluid, predominantly on the right side. Therapeutic thoracentesis was performed.
Given this reaccumulation of the exudative pleural effusion after completing a course of antibiotics, we suspected medication-induced fluid accumulation as the cause of the recurrent pleural effusion and possible small pericardial effusion in the echocardiogram. The patient was on sirolimus, acyclovir, and dasatinib. Although any of these drugs could potentially cause pleural effusion, we decided to stop dasatinib first because patients treated with dasatinib had the highest incidence of pleural effusion. Since the patient was in remission with no
The patient was discharged from the hospital without requiring oxygen, and his chest X-ray on discharge date showed no signs of pleural effusion (Figure 4). A repeat echocardiogram 2 weeks later upon the patient’s follow-up with his primary oncologist showed trivial pericardial effusion. Given the undetectable
Discussion
Pleural effusion following bone marrow transplantation can be classified as early onset (less than 100 days following HSCT) or late onset (more than 100 days following HSCT) effusion (Figure 5). Infections and volume overload can cause pleural effusion in either period, but they are more common with early-onset pleural effusion [4]. Engraftment syndrome is more commonly associated with early-onset pleural effusion, whereas serositis caused by GVHD is more common in late-onset pleural effusion. Fluid overload is the most common noninfectious cause of pleural effusion [4]. Chronic GVHD can manifest as a polyserositis-like pericardial effusion with pleural effusion and ascites [4,5]. Hepatic veno-occlusive disease, a clinical syndrome that can occur in patients who have undergone chemotherapy and radiation therapy, is characterized by jaundice, abdominal pain, and hyperbilirubinemia, and it can be complicated by multiorgan failure and a polyserositis-like pleural effusion [4,6]. Malignant pleural effusion is rare after HSCT, but it can be seen in refractory non-Hodgkin’s lymphoma [4].
Drug-related pleural disease is a rare cause of pleural effusion, in general, and it has not been reported in patients who have undergone HSCT until now. Many medications can cause exudative pleural effusions, with the exception of cardiovascular medications, which can cause transudative pleural effusions [7]. The possible mechanisms of drug-related pleural disease could be a direct toxic effect, allergic reaction, or chemical inflammation [7,8]. Drug-induced effusions are usually bilateral and dose related. Some medications such as valproic acid can cause an eosinophilic pleural effusion, and other medications such as dasatinib can cause lymphocytosis. Most drug-induced pleural effusions resolve after the causative medication is stopped [7,8].
Several medications that can cause pleural effusion in HCST patients such as mammalian target of rapamycin (mTOR) inhibitors (eg, sirolimus) to prevent GVHD [7,8] or mycophenolate or tyrosine kinase inhibitors (eg, dasatinib).
Our patient was taking 3 medications that can potentially cause pleural effusion: acyclovir, sirolimus, and dasatinib [7,8]. Fluid accumulation-like pleural effusion has been described as a possible adverse effect of mTOR inhibitors such as everolimus and sirolimus. The possible mechanisms include inhibition of lymphangiogenesis by inhibiting vascular endothelial growth factor, which is responsible for endothelial lymphatic proliferation [9].
There has been 1 case of a patient treated with acyclovir who presented with fever and, upon chest X-ray, showed pleural effusion and pulmonary infiltrates [10]. The cause of the condition was diagnosed by exclusion of infections and pulmonary embolism, and the pleural effusion resolved after acyclovir was stopped.
Dasatinib is a tyrosine kinase inhibitor that is used to treat CML and Philadelphia chromosome-positive ALL to prevent relapse [3]. Dasatinib typically causes exudative pleural effusion similar to that caused by infections and can therefore be missed. A common reaction to dasatinib is fluid accumulation, which may present as pleural effusion, pericardial effusion, or ascites [11]. Pleural effusion is found in 10–35% of CML patients taking dasatinib, and the risk increases with high-dose and high-frequency administration [12]. Dasatinib-associated pleural effusion is typically exudative with lymphocytic predominance [13]. Chylothorax is a less common complication of dasatinib therapy [14].
The mechanism of dasatinib-induced pleural effusion is unclear, but it is related to the immune system based on observational studies of patients with large granular lymphocytosis, both in the peripheral blood and in the pleural effusion, which were identified as natural killer cells and cytotoxic T cells [15]. However, these observational studies were conducted on patients with CML and not on patients who had undergone HSCT. Another possible pathophysiological mechanism could be secondary to tyrosine kinase inhibitors inhibiting platelet-derived growth factor, which is normally involved in angiogenesis and maintaining vessel integrity. Leaky vessels could be a possible histological mechanism underlying fluid accumulation [16].
The risk factors for dasatinib-induced pleural effusion in CML have been investigated. In general, the incidence of dasatinib-induced pleural effusion is higher in patients with a history of cardiac disease, pre-existing autoimmune disease, or a rash while on therapy [17]. We propose additional risk factors, as illustrated by patients on mTOR inhibitors and mycophenolate in addition to dasatinib following HSCT being at higher risk.
The main treatment options for effusions include dose reduction, temporary drug interruption, or switching to an alternative tyrosine kinase inhibitor [18]. Therapy withdrawal after pneumonia treatment is the primary recommendation in the context of recurrent pleural effusion. Therefore, in the present case, we decided to stop dasatinib, as it is associated with a higher incidence of pleural effusion than sirolimus. Additionally, we did not try to decrease the dose or use alternative tyro-sine kinase inhibitors such as imatinib or bosutinib, since the patient’s ALL was in remission with no
In our patient, the diagnosis of dasatinib-induced recurrent pleural effusion was suspected after we treated infections and excluded continuing infection and malignancy as possible causes by means of analysis of pleural fluid obtained through thoracentesis. The patient’s exudative lymphocyte-predominant pleural effusion was attributed to dasatinib; however, his triglyceride level in the pleural fluid was not tested to exclude chylothorax. We stopped dasatinib therapy because of recurrent pleural effusion. The patient’s condition improved significantly, and he was discharged without oxygen requirement.
Conclusions
Although pleural effusion is a known complication of allogenic HSCT, drug-induced pleural effusion in patients who have undergone HSCT has not been described previously. Dasatinib-induced pleural effusion in HCST patients may differ from the effusion that develops in CML patients based on the following 4 points. First, there is a wide range of causes of pleural effusion in HSCT compared with CML. Second, the risk of infection could be higher in HSCT than in CML; therefore, thoracentesis is strongly suggested for patients who have undergone HSCT to exclude infection. Third, closer monitoring may be needed when patients start on tyrosine kinase inhibitors following HSCT. Fourth, complete drug withdrawal rather than dose reduction or use of alternative tyrosine kinase inhibitors could be appropriate in HSCT patients if ALL is in remission. This case demonstrates that it is also important to consider drugs as a cause of recurrent effusion following HSCT. Physicians may not be aware of the potential for drug-induced pleural disease because new drugs are continuously being introduced and there is limited clinical experience or knowledge about their adverse effects. Therefore, accurate medication reconciliation by reviewing home medications and adverse effects can prevent patients with pleural disease from undergoing unnecessary procedures such as drainage using an indwelling pleural catheter and could minimize the duration of hospital stay after the common causes of pleural effusion have been eliminated.
Figures
References:
1.. Haider S, Durairajan N, Soubani AO, Noninfectious pulmonary complications of haematopoietic stem cell transplantation: Eur Respir Rev, 2020; 29(156); 190119
2.. Krauth MT, Herndlhofer S, Schmook MT, Extensive pleural and pericardial effusion in chronic myeloid leukemia during treatment with dasatinib at 100 mg or 50 mg daily: Haematologica, 2011; 96; 163-66
3.. Watanabe A, Chansu S, Ogawa A, Prophylactic post-transplant dasatinib administration in a pediatric patient with Philadelphia chromosome-positive acute lymphoblastic leukemia: Pediatr Int, 2013; 55; e56-58
4.. Modi D, Jang H, Kim S, Incidence, etiology, and outcome of pleural effusions in allogeneic hematopoietic stem cell transplantation: Am J Hematol, 2016; 91; E341-47
5.. Leonard JT, Newell LF, Meyers G, Chronic GvHD-associated serositis and pericarditis: Bone Marrow Transplant, 2015; 50; 1098-104
6.. Bonifazi F, Barbato F, Ravaioli F, Diagnosis and treatment of VOD/SOS after allogeneic hematopoietic stem cell transplantation: Front Immunol, 2020; 11; 489
7.. Huggins JT, Sahn SA, Drug-induced pleural disease: Clin Chest Med, 2004; 25; 141-53
8.. Goldblatt M, Huggins JT, Doelken P, Dasatinib-induced pleural effusions: A lymphatic network disorder?: Am J Med Sci, 2009; 338; 414-17
9.. Pusateri DW, Muder RR, Fever, pulmonary infiltrates, and pleural effusion following acyclovir therapy for herpes zoster ophthalmicus: Chest, 1990; 98; 754-56
10.. Kim K, Jeong DW, Lee YH, Everolimus-induced systemic serositis after simultaneous liver and kidney transplantation: A case report: Transplant Proc, 2017; 49; 181-84
11.. Zhang ZC, Qaio JH, Ai HS, [A case report of refractory pleural effusion associated with dasatinib in the treatment of chronic myeloid leukemia]: Zhongguo Shi Yan Xue Ye Xue Za Zhi, 2015; 23; 401-6 [in Chinese]
12.. Masiello D, Gorospe G, Yang AS, The occurrence and management of fluid retention associated with TKI therapy in CML, with a focus on dasatinib: J Hematol Oncol, 2009; 2; 46
13.. Brixey AG, Light RW, Pleural effusions due to dasatinib: Curr Opin Pulm Med, 2010; 16(4); 351-56
14.. Huang YM, Wang CH, Huang JS, Dasatinib-related chylothorax: Turk J Hematol, 2015; 32; 68-72
15.. Schiffer CA, Cortes JE, Hochhaus A, Lymphocytosis after treatment with dasatinib in chronic myeloid leukemia: Effects on response and toxicity: Cancer, 2016; 122; 1398-407
16.. Quintás-Cardama A, Kantarjian H, O’Brien S, Pleural effusion in patients with chronic myelogenous leukemia treated with dasatinib after imatinib failure: J Clin Oncol, 2007; 25; 3908-14
17.. de Lavallade H, Punnialingam S, Milojkovic D, Pleural effusions in patients with chronic myeloid leukaemia treated with dasatinib may have an immune-mediated pathogenesis: Br J Haematol, 2008; 141; 745-47
18.. Shah NP, Kantarjian HM, Kim DW, Intermittent target inhibition with dasatinib 100 mg once daily preserves efficacy and improves tolerability in imatinib-resistant and -intolerant chronic-phase chronic myeloid leukemia: J Clin Oncol, 2008; 26; 3204-12
Figures
In Press
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943118
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942826
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942770
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943214
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250