10 September 2021: Articles 
Diffuse Lymphadenopathy Syndrome as a Flare-Up Manifestation in Lupus and Mixed Connective Tissue Disease Following Mild COVID-19
Challenging differential diagnosis, Unusual or unexpected effect of treatment, Rare coexistence of disease or pathology
Claudio Karsulovic12ABCDEFG*, Lia P. Hojman3ACDEF, Daniela L. Seelmann1ABCFG, Pamela A. Wurmann1ABCDEFGDOI: 10.12659/AJCR.932751
Am J Case Rep 2021; 22:e932751
Abstract
BACKGROUND: Manifestations of Coronavirus disease 2019 (COVID-19), caused by the SARS-CoV-2 virus, are highly variable among healthy populations. In connective tissue disease patients, the spectrum of clinical manifestations is even broader. In mild COVID-19 patients, diffuse lymphadenopathy (DL) has not been described as a late manifestation, and only severe COVID-19 has been associated with lupus flare-ups. Herein, we report 3 cases of connective tissue disease patients that presented with DL after diagnosis and complete resolution of mild COVID-19 disease.
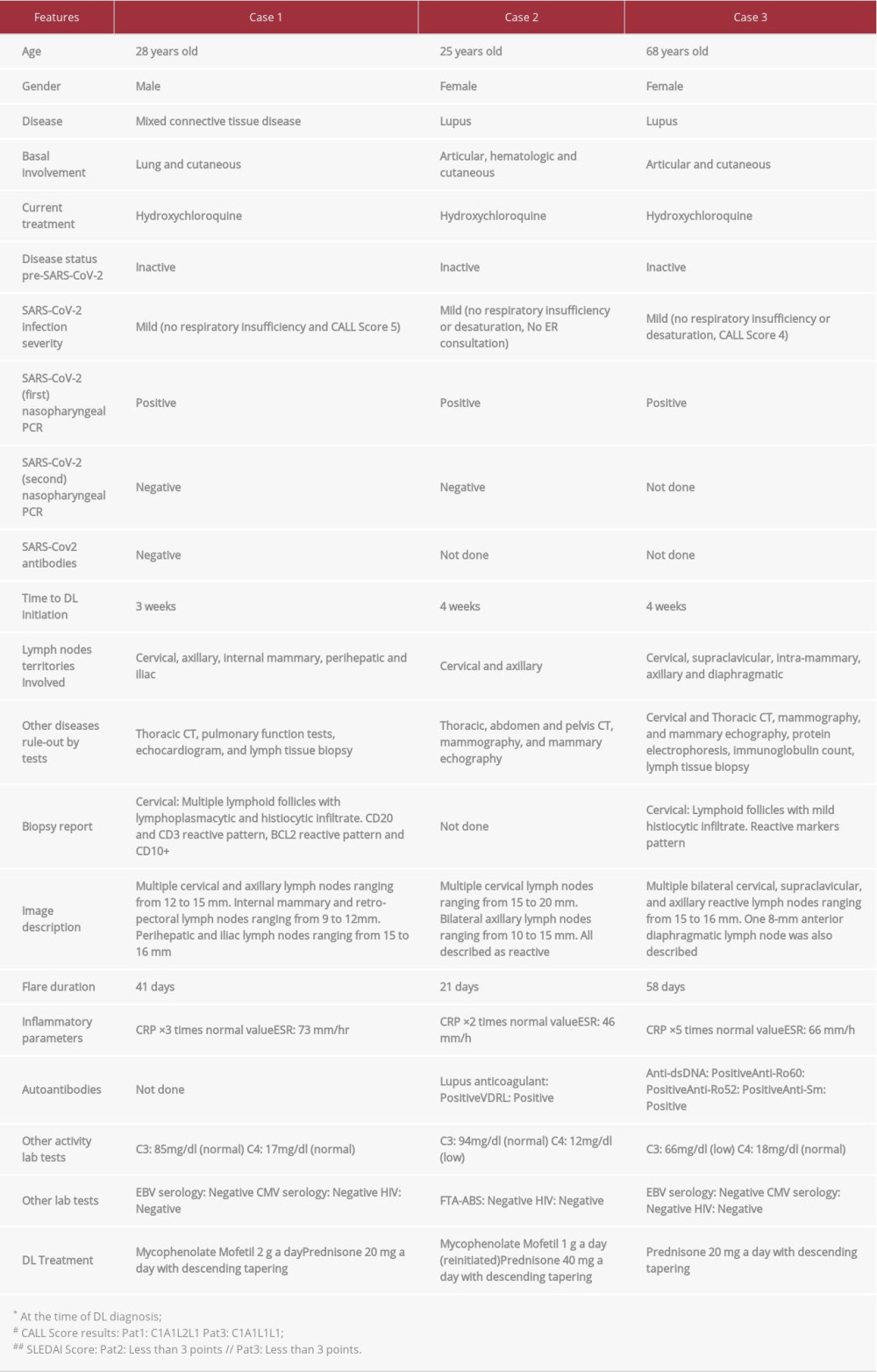
CASE REPORT: Case 1. A 28-year-old man with inactive lupus, mixed connective tissue disease (MCTD), and a history of lung and cutaneous involvement. He presented with fever, polyarthralgia, and multiple lymphadenopathies 3 weeks after COVID-19 disease resolution. After evaluation, immunosuppressive treatment was initiated, with rapid response. Case 2. A 25-year-old woman with inactive lupus with a history of articular, hematologic, and cutaneous involvement. Four weeks after resolution of COVID-19 disease, she presented with malaise and cervical lymphadenopathies. After laboratory testing and imaging, she was treated for lupus flare-up, with rapid response. Case 3. A 68-year-old woman with inactive lupus with a history of articular and cutaneous involvement. Four weeks after COVID-19 resolution, she presented with malaise and cervical and axillary lymphadenopathies. After extensive evaluation, immunosuppressive treatment resulted in a rapid response.
CONCLUSIONS: After 3 to 4 weeks of mild, outpatient-treated COVID-19 and complete resolution of symptoms, 3 patients with connective tissue disease presented diffuse lymphadenopathy associated with inflammatory and constitutional symptoms. Infectious and neoplastic causes were thoroughly ruled out. All patients responded to reintroduction of or an increase in immunosuppressive therapy. We recommend considering the diffuse lymphadenopathy as a possible post-acute COVID-19 syndrome (PACS) manifestation in these patients, mainly when they are in the inactive phase.
Keywords: COVID-19, Lupus Erythematosus, Systemic, mixed connective tissue disease, Symptom Flare Up, AIDS-Related Complex, COVID-19, Female, Humans, SARS-CoV-2
Background
COVID-19 manifestations are polymorphic and highly variable, both clinically and radiologically. Clinical presentations include DL involving cervical, subcarinal, and superior-mediastinal lymph nodes, mainly in severe COVID-19 cases [1,2]. Other reports show multiple cervical lymphadenopathies during the symptomatic phase of the disease [3,4]. To the best of our knowledge, there are no reports of persistent DL after the symptomatic phase of the disease.
Viral infections have been associated with connective tissue diseases (CTD) as a trigger at presentation and as clinical flare-up inductors. Herpes family viruses, like Epstein-Barr Virus and Cytomegalovirus, are the most frequently described agents, probably due to multiple immunologic features that favor a self-reactive response [5,6].
In COVID-19 disease, reports focus on ACE2 (angiotensin-converting enzyme 2), which acts as a receptor to the Spike protein from the SARS-CoV-2 virus [7].
Similar to patients with high cardiovascular and metabolic risk, lupus patients have specific genetic and epigenetic characteristics in ACE2 protein that can lead to a higher risk of infection and worse respiratory involvement [8].
In terms of progression, CTD patients have an enhanced IFN-γ response, leading to a more efficient viral infection control [9]. Nevertheless, inflammasome activation can be associated with a more aggressive late inflammatory response [10].
Several cases of flare-ups in previously stable CTD patients following COVID-19 have been described, most of them lupus patients who evolve with clinical and laboratory elements of a flare-up after severe COVID-19 disease. These flare-ups are usually severe and life-threatening, involving significant organ damage, with hematological, neurological, and macrophage-mediated systemic inflammatory response [11,12]. To the best of our knowledge, there is limited information about clinical presentation or course of mild flare-ups in CTD diseases. Here, we present 3 cases with DL from September to November 2020 at the Rheumatology Section of Hospital Clinico de la Universidad de Chile. Two patients with inactive lupus previous to COVID-19 disease and 1 with MCTD (also inactive) were diagnosed and treated as flare-ups after thorough laboratory and imaging studies.
Case Reports
CASE 1:
A 28-year-old man with MCTD with lung and cutaneous involvement (meeting Alarcón-Segovia criteria of anti-RNP titers over 1: 1000 plus synovitis, Raynaud’s, and hand edema), diagnosed with mild COVID-19. He presented with low-grade fever, sore throat, dry cough, and myalgias, and received outpatient care. After 3 weeks, he reconsulted with fevers, weight loss, cervical lymphadenopathies, and polyarthralgia. The patient underwent extensive studies, including pulmonary function tests, echo-cardiogram, thoracic CT, and cervical lymph node biopsy; the latter was regarded as a cervical reactive lymph node. After 3 weeks of exhaustive study and persistent DL, flare-up treatment was initiated using prednisone 20 mg and mycopheno-late mofetil 2 g a day. Seven-day follow-up showed satisfactory recovery (Table 1).
CASE 2:
A 25-year-old woman with inactive lupus with articular, hematologic, and cutaneous involvement (meeting ACR/EULAR criteria with ANA titers over 1: 320, thrombocytopenia, synovitis, and non-scarring alopecia) was diagnosed with mild COVID-19. She presented with fever, myalgia, and asthenia, and received outpatient care. Four weeks after resolution of COVID-19, she began to have headaches and bilateral axillar pain with palpable lymph nodes. Evaluation included thoracic, abdomen, and pelvis CT, mammography, breast ultrasound, and laboratory tests. Thoracic CT images showed multiple lymphadenopathies in cervical and axillar territories.
Laboratory testing showed that C4 dropped from 21 mg/dl to 12 mg/dl. After 3 more weeks of persistent DL, mycopheno-late mofetil was reinitiated (following 9 months of withdrawal), using 500 mg every 12 h. In addition, prednisone 40 mg a day was also initiated with tapering over a 2-month period (Table 1).
CASE 3:
A 68-year-old woman with previously inactive lupus with articular and cutaneous involvement (meeting ACR/EULAR criteria with ANA titers over 1: 640, synovitis, non-scarring alopecia, and hypocomplementemia) was diagnosed with mild COVID-19. She presented with low-grade fever, asthenia, and dry cough, and was treated at home. Four weeks after a positive SARS-CoV-2 polymerase chain reaction test, she presented with multiple cervical and axillar lymphadenopathies along with persistent asthenia. The patient underwent extensive evaluation, including thoracic and cervical CT, mammography, and breast ultrasound. Bilateral cervical, supraclavicular, and axillary lymphadenopathies were found. Biopsy showed lymphoid follicles with mild histiocytic infiltrate. Laboratory testing included protein electrophoresis, complement levels, and anti-DNAds. The latter was highly positive, along with a decrease of C3 to pre-infection values. Prednisone 20 mg was initiated after 6 weeks of DL, with a satisfactory response (Table 1).
Discussion
In COVID-19 disease, cervical and intrathoracic lymphadenopathies have been described during the symptomatic phase. Delayed initiation of DL with the involvement of deep areas and lasting several weeks, and after a mild clinical course of the disease and complete resolution of viral symptoms, has never been described before and poses the question of whether this could be a different pathological entity.
Lymphadenopathy is uncommon as an isolated manifestation in lupus and MCTD flare-ups [13], and the present case series is the first to report on patients presenting with this clinical manifestation. Neoplastic and other inflammatory diseases were thoroughly ruled out, and specific features, such as falling complement levels, were demonstrated in 2 of them.
Kikuchi-Fujimoto disease (non-infectious inflammatory lymph-adenopathy) was specially ruled out since it is one of the primary differential diagnoses in these cases. This condition is diagnosed mainly by lymph node biopsy, which typically shows non-suppurative, non-caseating necrosis [14]. In 2 of our patients with lymph node biopsy, images and histology did not show characteristic necrosis and related features. In patient 2, for whom a biopsy was not performed, Kikuchi-Fujimoto disease could not be ruled out; nevertheless, thoracic and cervical CT scan images did not find typical features, and central nodal necrotic changes are only seen in 20% of cases.
COVID-19 been associated with macrophage activation syndrome and hemophagocytic lymphohistiocytosis during the acute phase [15]. Macrophages and circulating monocytes are essential for T and B lymphocyte recruiting and activation in lymph tissue [16]. During COVID-19 disease, circulating monocytes become activated and express the same inflammatory phenotype markers described in lupus patients, even during periods of clinical inactivity [17,18]. In a lupus patient with clinical or subclinical activity, inflammatory-expressing monocytes/macrophages have an enhanced inflammasome activation, which needs 2 different stimuli to occur: production of IL-1β and IL18 [19]. It is reasonable to hypothesize that COVID-19 could represent a sufficient “second hit” to stimulate lymph node-recruited monocytes, showing a clinical diffuse lymphadenopathy disease with a persistent local inflammatory reaction.
The T helper type 1 lymphocytes and macrophage-mediated response to the infection and, as recently reported, to (mRNA)-based SARS-CoV-2 vaccines, can trigger cutaneous lupus flare-ups. However, it has not been reported in systemic lupus, neither with mRNA vaccines nor inactivated virus-based ones [20]. Our patients were not vaccinated at the time they were treated and studied.
Activation of complement cascades due to COVID-19, resulting in complement decrease levels, has been reported, but mainly in severe presentation of the disease and in pneumonia animal models [21]. Since our patients had mild COVID-19 disease, we did not attribute it to COVID-19.
We want to highlight 4 critical points of post-COVID-19 DL that support the theory presented herein: (1) Three previously inactive patients presented with DL after 3 to 4 weeks of mild COVID-19 disease, and DL was an unusual presentation and had appeared for the first time in these patients; (2) Imaging and laboratory study without other possible explanations for these features, with an increase in basal autoantibodies titers and complement levels; (3) DL after 3 to 4 weeks of symptom onset and lasting several weeks in all 3 patients with satisfactory and rapid recovery upon glucocorticoid/immunosuppressive treatment initiation, and (4) A plausible pathogenic explanation for this clinical observation.
Conclusions
In previously inactive CTD patients, viral disease could be a trigger of a flare-up of the disease. In lupus patients, even when diffuse lymphadenopathy is uncommon as an isolated manifestation of a flare-up of the disease, we are increasingly treating patients with these symptoms after complete resolution of mild COVID-19. Avoiding expensive and time-consuming studies, we recommend considering that COVID-19 can trigger disease flare-ups in which, in selected cases, DL could be a manifestation of a lupus flare-up with or without other clinical features.
References:
1.. Valette X, du Cheyron D, Goursaud S, Mediastinal lymphadenopathy in patients with severe COVID-19: Lancet Infect Dis, 2020; 20(11); 1230
2.. Mughal MS, Rehman R, Osman R, Hilar lymphadenopathy, a novel finding in the setting of coronavirus disease (COVID-19): A case report: J Med Case Rep, 2020; 14(1); 124
3.. Distinguin L, Ammar A, Lechien JR, MRI of patients infected with COVID-19 revealed cervical lymphadenopathy: Ear Nose Throat J, 2021; 100(1); 26-28
4.. Haigh K, Syrimi ZJ, Irvine S, Hyperinflammation with COVID-19: The key to patient deterioration?: Clin Infect Pract, 2020; 7; 100033
5.. Meyer O, Kahn MF, Grossin M, Parvovirus B19 infection can induce histiocytic necrotizing lymphadenitis (Kikuchi’s disease) associated with systemic lupus erythematosus: Lupus, 1991; 1(1); 37-41
6.. Jung J-Y, Suh C-H, Infection in systemic lupus erythematosus, similarities, and differences with lupus flare: Korean J Intern Med, 2017; 32(3); 429-38
7.. Tariq S, Van Eeden C, Tervaert JWC, Osman MS, COVID-19, rheumatic diseases and immune dysregulation – a perspective: Clin Rheumatol, 2021; 40(2); 433-42
8.. Sawalha AH, Zhao M, Coit P, Lu Q, Epigenetic dysregulation of ACE2 and interferon-regulated genes might suggest increased COVID-19 susceptibility and severity in lupus patients: Clin Immunol, 2020; 215; 108410
9.. Muskardin TLW, Niewold TB, Type I interferon in rheumatic diseases: Nat Rev Rheumatol, 2018; 14(4); 214-28
10.. Hadjadj J, Yatim N, Barnabei L, Impaired type I interferon activity and inflammatory responses in severe COVID-19 patients: Science, 2020; 369(6504); 718-24
11.. Raghavan S, Gonakoti S, Asemota IR, Mba B, A Case of systemic lupus erythematosus flare triggered by severe coronavirus disease 2019: J Clin Rheumatol, 2020; 26(6); 234-35
12.. Katz-Agranov N, Zandman-Goddard G, Autoimmunity and COVID-19 –the microbiotal connection: Autoimmun Rev, 2021; 20(8); 102865
13.. Adamichou C, Bertsias G, Flares in systemic lupus erythematosus: Diagnosis, risk factors and preventive strategies: Mediterr J Rheumatol, 2017; 28(1); 4-12
14.. Alsolami A, Altirkistani R, Tayeb S, A case of recurrent cervical lymphadenopathy due to Kikuchi-Fujimoto disease: Am J Case Rep, 2021; 22; e928760
15.. Karsulovic C, Lopez M, Tempio F, mTORC inhibitor Sirolimus depro-grams monocytes in “cytokine storm” in SARS-CoV2 secondary hemophagocytic lymphohistiocytosis – like syndrome: Clin Immunol, 2020; 218; 108539
16.. Crispin JC, Labonte AC, Kegerreis B, Identification of alterations in macrophage activation associated with disease activity in systemic lupus erythematosus: PLoS One, 2018; 13(12); e0208132
17.. Liu A-C, Yang Y, Li M-T, Macrophage activation syndrome in systemic lupus erythematosus: a multicenter, case-control study in China: Clin Rheumatol, 2017; 37(1); 93-100
18.. Hirose S, Lin Q, Ohtsuji M, Nishimura H, Verbeek JS, Monocyte subsets involved in the development of systemic lupus erythematosus and rheumatoid arthritis: Int Immunol, 2019; 31(11); 687-96
19.. Kahlenberg JM, Carmona-Rivera C, Smith CK, Kaplan MJ, Neutrophil extra-cellular trap – associated protein activation of the NLRP3 inflammasome is enhanced in lupus macrophages: J Immunol, 2013; 190(3); 1217-26
20.. Niebel D, Ralser-Isselstein V, Jaschke K, Exacerbation of subacute cutaneous lupus erythematosus following vaccination with BNT162b2 mRNA vaccine: Dermatol Ther, 2021; 34(4); e15017
21.. Wang X, Sahu KK, Cerny J, Coagulopathy, endothelial dysfunction, thrombotic microangiopathy and complement activation: Potential role of complement system inhibition in COVID-19: J Thromb Thrombolysis, 2021; 51(3); 657-62
In Press
06 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942937
12 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943244
13 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943275
13 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943411
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250