13 November 2021: Articles 
Sclerosing Angiomatoid Nodular Transformation (SANT): A Rare Splenic Tumor and Unusual Cause of Anemia
Challenging differential diagnosis, Unusual or unexpected effect of treatment, Rare disease
Bee Shan Ong1EF*, Rebecca Thomas1ABDEFDOI: 10.12659/AJCR.933598
Am J Case Rep 2021; 22:e933598
Abstract
BACKGROUND: Sclerosing angiomatoid nodular transformation (SANT) of the spleen is a rare benign disease of the splenic red pulp of unknown etiology. Definite diagnosis is made on histopathology alone as it has no diagnostic radiologic characteristics.
CASE REPORT: We present a case of a large incidental splenic mass in a middle-aged man, whose refractory anemia resolved with splenectomy. Our initial imaging studies failed to differentiate this lesion from other splenic lesions like hamartoma and haemangioma. To the best of our knowledge, no SANT cases in the literature have been investigated with red cell scan, as performed in our patient, nor has any case had resolved anemia after treatment. Splenectomy was offered as malignancy could not be safely excluded. Histopathology confirmed the diagnosis of SANT. The patient made good recovery and had no signs of recurrence 2 years later.
CONCLUSIONS: SANT of the spleen is a rare condition that can mimic many other splenic tumors and poses diagnostic challenge when histopathology is unavailable. Our case adds to the number of SANT reported in literature in the hope of elucidating the pathophysiology of this rare condition.
Keywords: Anemia, Immunohistochemistry, Nuclear Medicine, Histiocytoma, Benign Fibrous, Humans, Male, Splenic Diseases, Splenic Neoplasms
Background
Sclerosing angiomatoid nodular transformation (SANT) of the spleen is a rare benign vascular disease originating from splenic red pulp. Most patients are asymptomatic, with incidental findings of splenomegaly on physical examination or imaging performed for other unrelated conditions, as in our patient. Preoperative diagnosis remains a challenge as there is currently no biomarker specific for SANT. Imaging plays an important role in aiding diagnosis, although there is no pathognomonic imaging feature. Due to the diagnostic dilemma, most patients are offered splenectomy due to the risk of splenic rupture and malignancy. We herein report a case of SANT that was initially thought to be a hemangioma or hamartoma, and the patient was offered splenectomy with associated resolution of chronic intractable anemia.
A literature search was performed to understand the clinicopathologic manifestations and differential diagnoses of SANT.
Case Report
A 57-year-old man presented with an incidental large splenic mass on ultrasound performed for investigating a sensation of incomplete micturition. His background medical history was significant for chronic anemia refractory to iron supplementation and dietary modifications for almost 8 years. His anemia was investigated with a panendoscopy, which was unremarkable. Other past medical history included gastroesophageal reflux disease, hypertension, and chronic schizophrenia. Medications included nifedipine, olanzapine, ranitidine, and iron tablets. Surgically, he had bilateral inguinal hernias repair at the age of 5 and right hip surgery in his teens. On physical examination, his abdomen was soft and non-tender with no organomegaly.
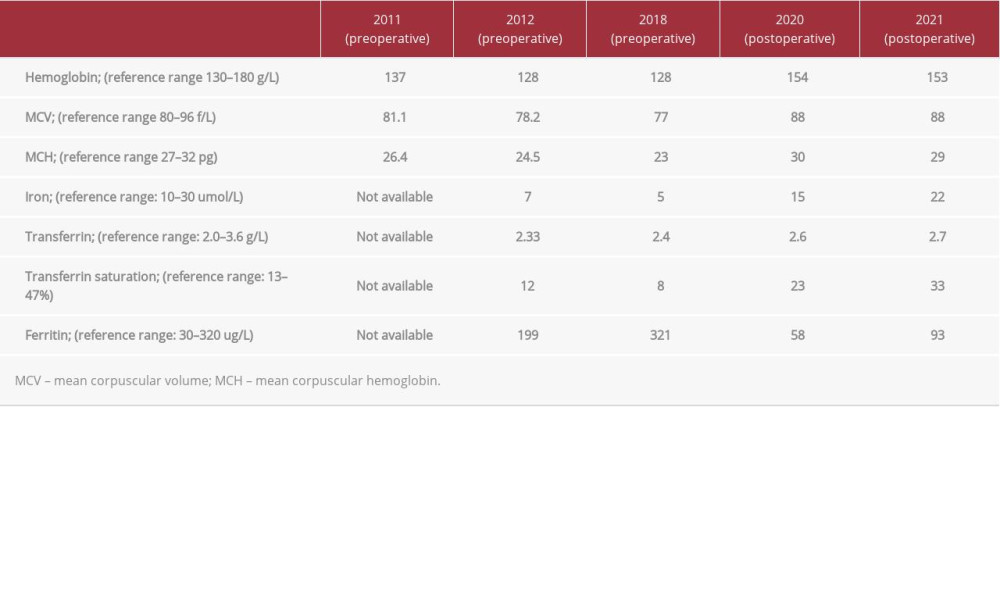
Blood tests were unremarkable except for microcytic hypochromic anemia and a mildly raised C reactive protein of 46.7 mg/L (upper limit reference limit: 8 mg/L). Table 1 compares the patient’s hemoglobin and iron studies level during the pre- and postoperative periods (he was operated on in 2019).
A CT abdomen/pelvis revealed a relatively well-circumscribed 8×10×6 cm homogeneous mass centered within the superior pole of the spleen containing several punctate calcifications scattered around the periphery, which favored a benign lesion suggestive of hemangioma (Figure 1).
MRI scan (Figures 2, 3) showed a 104×87 mm solitary upper-pole splenic lesion with extensive signal dropout which appeared primarily related to hemosiderin deposition and to a lesser extent calcification. The lesion was hypoenhancing relative to the splenic parenchyma in the arterial phase, becoming more isointense on the portal venous phase and remaining so in the delayed phase. On T2 signal, the lesion was isointense to the spleen and minimally hyperintense relative to the liver. This was atypical for hemangioma.
A red cell scan showed an absent perfusion in the lesion initially with only a thin rim of patchy tracer activity at the periphery. There was minor filling within the body of the lesion on delayed images. This was non-diagnostic for a splenic hemangioma but raised the possibility of a splenic hamartoma (Figure 4).
A PET-FDG scan was done to rule out splenic lymphoma. It showed mild heterogenous FDG uptake (SUV-max 5.4 [standardized uptake value]) of the splenic mass and no FDG avid lymphadenopathy (Figure 5).
The decision to operate was made given the risk of rupture and uncertainty in diagnosis as malignancy could not be safely excluded. Core biopsy was not attempted due to the fear of seeding (in the case of malignancy) and bleeding. The patient underwent open splenectomy after having the required vaccination as per national guidelines. Intraoperatively, there was a 10-cm multilobulated firm lesion at the superolateral aspect of spleen. The pancreatic tail was seen and preserved.
Macroscopically, the entire specimen weighed 430 g. The hilum appeared free of the lesion. The cut surface revealed a sclerosed, firm trabeculated lesion which appeared largely encapsulated, with a red-to-tan cut surface, with the largest dimension 65×80 mm on the cut surface. No lymph nodes were macroscopically identified within the hilar fat (Figure 6).
Microscopically, there was a relatively well-circumscribed lesion within the splenic parenchyma with areas of nodular architecture. Extensive deposition of sclerotic collagen was noted centrally, and numerous small vessels lined by bland endothelium were present, the majority with compressed lumina. Extravasation of red cells was evident, and there was hemosiderin deposition in areas. The background stroma included areas containing bland spindle cells, and a mixed inflammatory cell infiltrate dominated by histiocytes (Figure 7). Immunohistochemistry (Figure 8) demonstrated that the capillary vessels were positive for CD31 and CD34 but negative for CD 8; the sinusoids were positive for CD31 and CD8 but CD34 negative; and only CD31 was positive in the small veins. Macrophages were positive for CD68. Overall, the morphology and immunophenotype were consistent with benign sclerosing angiomatoid nodular transformation.
The patient had an uneventful postoperative recovery, with immediate resolution of his recalcitrant anemia and he remained well at 24 months.
Discussion
Sclerosing angiomatoid nodular transformation (SANT) of the spleen is an uncommon benign vascular disease originating from splenic red pulp [1]. Initially thought to be a hamartoma or hemangioma by some authors, Martel first introduced the term SANT in 2004 after examination of a series of 25 cases [2].
Epidemiologically, it occurs predominantly in middle-aged people, with female pre-dominance [3,4]. The etiology of SANT is unclear, and it can present as solitary or multiple lesions. While some studies hypothesize that SANT is a response to hamartoma and trauma, others demonstrated a relationship with Epstein-Barr virus infection (hence an inflammatory pseudotumor) and immunoglobulin G4-related autoimmune diseases [5–7].
Most patients with SANT are asymptomatic, with incidental findings of splenomegaly on physical examination or imaging performed for other unrelated conditions [4,8], as occurred in our patient, but some patients present with non-specific symptoms of abdominal pain, nausea, vomiting, anemia or malnutrition [5,9,10]. Our patient had chronic anemia not responding to dietary changes and iron supplementation. More detailed analysis of patient’s blood was not possible due to rapid resolution of anemia following splenectomy, which was an unexpected and serendipitous outcome. Our case appears to be the first ever reported in the literature of resolution of chronic anemia following splenectomy for SANT. Therefore, the exact pathophysiology is unknown but we hypothesize that any tumor in the spleen increases red blood cells destruction, as evident by histopathology showing extravasation of red blood cells. It is also possible that it was an anemia of chronic disease, which is a chronic functional iron-deficiency anemia on a background of chronic disease in this evolving splenic tumor [11].
While SANT had been found in the setting of some malignancies, the lesion itself is benign, with no recurrence or malignant potential reported to date [12,13]. The diagnosis of SANT can be challenging without histological confirmation. As there is currently no biomarker specific for SANT, imaging plays an important role in aiding diagnosis.
The presence of internal vascularity on color Doppler ultra-sound in a spleen with heterogenous echotexture may provide some clues suggesting SANT. However, ultrasound alone is insufficient, as SANT can have different appearances on an ultrasound [14].
In 2021, Liao et al studied 18 patients with SANT and identified computed tomography (CT) and magnetic resonance imaging (MRI) features of this condition. These features are: (a) clear boundary with smooth margin or lobulation, (b) occasional calcification and mild hypoattenuation on plain CT, (c) heterogenous hypointensity on T2-weighted imaging or diffusion-weighted imaging, (d) reduced local signal intensity on in-phase images in comparison to out-of-phase images, (e) central fibrous scar, and (f) enhancement characteristics. The latter include progressive and centripetal, nodular, or delayed central fibrous scar enhancement as well as spoke-wheel pattern. Spoke-wheel pattern correlates with histopathology, in which angiomatous nodules concentrate around the periphery of the lesion, in addition to a radiating pattern between fibrous scar branches [15].
These imaging features are, however, only for solitary SANT. The imaging features of multiple-lesion SANT are different, especially on T1- and T2-weighted imaging and contrast-enhanced imaging. It was found that multiple SANT has inconsistent imaging findings, and this may be due to a smaller number of cases reported or differing pathogenesis between single and multiple SANTs [16]. More recently, a study of 5 patients with SANT by Shao et al proposed the finding of peripheral enhancement of a splenic lesion in arterial phase CT to be considered a sign suggestive of SANT [17].
In positron emission tomography-computed tomography (PET-CT) scans, SANT demonstrates a low degree of FDG up-take, which can be explained by the number of hemosiderin-laden macrophages and inflammatory cells in angiomatoid nodules [18]. This characteristic of SANT makes it difficult to rule out malignancy. In a study by Matsubara, a patient with SANT was followed up with PET-CT 5 years after an initial diagnosis, with the subsequent scan showing an increase in FDG uptake as compared to the first scan [19]. The patient subsequently had splenectomy even though the size of the spleen had reduced.
SANT has wide differential diagnoses ranging from benign conditions such as angioma, hamartoma, lymphangioma, littoral cell angioma, and inflammatory pseudotumor to malignant conditions like angiosarcoma, lymphoma, and metastatic splenic tumor. Despite efforts to distinguish SANT from other differentials, diagnosing SANT solely from imaging is challenging [5].
In general, splenectomy remains the first-line management for SANT. This avoids the risk of suspicion of malignancy and spontaneous rupture of the spleen, although to date, only 1 case of SANT had ruptured, as reported by Pelizzo et al in a 9-week-old female infant [20]. Most patients underwent open or laparoscopic splenectomy. The operative time, blood loss, and postoperative complications were comparable between the 2 approaches. However, shorter hospital stay was seen with the laparoscopic group. Other factor for consideration when choosing either an open or laparoscopic approach would be the size of the spleen on preoperative imaging, as a massive spleen can limit laparoscopic space. In addition, when the diagnosis of malignancy is unable to be safely excluded, it is important to preserve splenic integrity to achieve en bloc resections [21,22].
There were also studies on the option of obtaining a core needle biopsy of the splenic mass, but this is not recommended, as the result was low yield and it has potential complications of hemorrhage and damage to the bowel, lungs, and diaphragm [13].
In a recent retrospective study by Jin et al in 2020, the option of laparoscopic partial splenectomy (LPS) to treat SANT became promising as it avoids the risks of asplenia and overwhelming post-splenectomy infection (OPSI). In that study, LPS was successfully performed in 13 patients following intraoperative frozen section examination to exclude malignancy. None of the patients who underwent LPS had recurrence during the median follow-up period of 46 months, and the postoperative platelet count peak was significantly lower compared to open or laparoscopic groups [21].
The only method to confirm SANT is immunohistochemistry. It has characteristic features of CD34(−) CD31(+) CD8(+) for sinusoids, CD34(+) CD31(+) CD8(−) for capillaries, and CD34(−) CD31(+) CD8(−) for small veins. CD68 is positive in macrophages. Macroscopically, SANT presents as a well-demarcated mass with a central gray-white stellate scar and peripheral reddish-brown nodules. On a microscopic view, multiple angiomatous nodules surrounded by fibrous collagen tissue can be seen, in addition to lining of micronodular vascular spaces by plump endothelial cells interspersed with ovoid spindle cells [17,23].
Conclusions
Clinicians should be cognisant of SANT as an unusual cause of refractory anemia. It is a rare splenic lesion with unknown etiology, and immunohistochemistry is the only way to confirm the diagnosis. Splenectomy is the only treatment and prognosis is excellent. Further research should be encouraged to explore the link between anemia and SANT.
Figures
References:
1.. Krishnan J, Frizzera G, Two splenic lesions in need of clarification: Hamartoma and inflammatory pseudotumor: Semin Diagn Pathol, 2003; 20; 94-104
2.. Martel M, Cheuk W, Lombardi L, Sclerosing angiomatoid nodular transformation (SANT): Report of 25 cases of a distinctive benign splenic lesion: Am J Surg Pathol, 2004; 28; 1268-79
3.. Li L, Fisher DA, Stanek AE, Sclerosing angiomatoid nodular transformation (SANT) of the spleen: Addition of a case with focal CD68 staining and distinctive CT features: Am J Surg Pathol, 2005; 29; 839-41
4.. Vigorito R, Scaramuzza D, Pellegrinelli A, Marchianò A, Sclerosing angiomatoid nodular transformation (SANT) of the spleen: a case report on CT and MRI: BJR Case Rep, 2019; 5; 20180036
5.. Yoshii H, Izumi H, Nomi M, Sclerosing angiomatoid nodular transformation of the spleen: A case report: Tokai J Exp Clin Med, 2020; 45(4); 236-42
6.. Weinreb I, Bailey D, Battaglia D, CD30 and Epstein-Barr virus RNA expression in sclerosing angiomatoid nodular transformation of spleen: Virchows Arch, 2007; 451(1); 73-79
7.. Nagai Y, Hayama N, Kishimoto T, Predominance of IgG4+ plasma cells and CD68 positivity in sclerosing angiomatoid nodular transformation (SANT): Histopathology, 2008; 53(4); 495-98
8.. Ma J, Zhang W, Wang L, Imaging features of sclerosing angiomatoid nodular transformation in spleen: J Comput Assist Tomogr, 2019; 43; 863-69
9.. Pinheiro JL, Catarino S, Sclerosing angiomatoid nodular transformation of the spleen: Case report of a metastatic carcinoma-simulating disorder: J Surg Case Rep, 2019; 2019; rjz249
10.. Chikhladze S, Lederer AK, Fichtner-Feigl S, Sclerosing angiomatoid nodular transformation of the spleen, a rare cause for splenectomy: Two case reports: World J Clin Cases, 2020; 8; 103-9
11.. Thomas DW, Hinchliffe RF, Briggs C, Guideline for the laboratory diagnosis of functional iron deficiency: Br J Haematol, 2013; 161(5); 639-48
12.. Lee HJ, Choi SY, Huang SM, Sul JY, Kim JM, Sclerosing angiomatoid nodular transformation (SANT) in spleen: A case report: Korean J Pathol, 2011; 45; 111-14
13.. Aziret M, Yılmaz F, Kalpakçı Y, Sclerosing angiomatoid nodular transformation presenting with thrombocytopenia after laparoscopic splenectomy – case report and systematic review of 230 patients: Ann Med Surg (Lond), 2020; 60; 201-10
14.. Wang HL, Li KW, Wang J, Sclerosing angiomatoid nodular transformation of the spleen: Report of five cases and review of literature: Chin Med J, 2012; 125; 2386-89
15.. Karaosmanoglu DA, Karcaaltincaba M, Akata D, CT and MRI findings of scle-rosing angiomatoid nodular transformation of the spleen: A spoke wheel patter: Korean J Radiol, 2008(Suppl); S52-55
16.. Liao J, Wang Z, Li Q, CT and MRI features of sclerosing angiomatoid nodular transformation of the spleen: A report of 18 patients with pathologic correlation: Diagn Interv Imaging, 2021; 102(6); 389-96
17.. Shao H, Lu B, Shen Z, Liu F, Sclerosing angiomatoid nodular transformation of the spleen: Analysis of clinical and pathological features in five cases: Front Surg, 2021; 7; 609284
18.. Imamura Y, Nakajima R, Hatta K, Sclerosing angiomatoid nodular transformation (SANT) of the spleen: A case report with FDG-PET findings and literature review: Acta Radiol Open, 2016; 5(8); 2058460116649799
19.. Matsubara K, Oshita A, Nishisaka T, A case of sclerosing angiomatoid nodular transformation of the spleen with increased accumulation of fluorodeoxyglucose after 5-year follow-up: Int J Surg Case Rep, 2017; 39; 9-13
20.. Pelizzo G, Villanacci V, Lorenzi L, Sclerosing angiomatoid nodular transformation presenting with abdominal hemorrhage: First report in infancy: Pediatr Rep, 2019; 11(2); 7848
21.. Jin Y, Hu H, Regmi P, Li F, Cheng N, Treatment options for sclerosing angiomatoid nodular transformation of spleen: HPB (Oxford), 2020; 22(11); 1577-82
22.. Cipolla C, Florena AM, Ferrara G, Sclerosing angiomatoid nodular transformation: Laparoscopic splenectomy as therapeutic and diagnostic approach at the same time: Case Rep Surg, 2018; 2018; 7020538
23.. Hou J, Ji Y, Tan YS, [Sclerosing angiomatoid nodular transformation of spleen: A clinicopathologic study of 10 cases with review of literature]: Zhonghua Bing Li Xue Za Zhi, 2010; 39(2); 84-87 [in Chinese]
Figures
In Press
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943687
17 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943070
17 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943370
18 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943803
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250