19 November 2021: Articles 
Delayed Tentorial Subdural Hematoma Caused by Traumatic Posterior Cerebral Artery Aneurysm: A Case Report and Literature Review
Unusual clinical course
Michiyasu Fuga1ABCDEF*, Toshihide Tanaka1CDEF, Ryo Nogami1BE, Rintaro Tachi1BE, Akihiko Teshigawara1ABD, Toshihiro Ishibashi2ACDF, Yuzuru Hasegawa1DF, Yuichi Murayama2ACDFDOI: 10.12659/AJCR.933771
Am J Case Rep 2021; 22:e933771
Abstract
BACKGROUND: Subdural hematoma (SDH) caused by traumatic intracranial aneurysm (TICA) is rare. TICAs are known to rupture easily, resulting in a high morbidity and mortality rate. Therefore, accurate diagnosis and treatment are crucial for preserving life. We describe a case of delayed SDH in the setting of posterior cerebral artery (PCA) aneurysm.
CASE REPORT: A 42-year-old man presented with sustained head injury from a traffic accident, and was being followed-up conservatively for traumatic SDH and subarachnoid hemorrhage. Three weeks after the head trauma, the patient developed a sudden deterioration of mental status and disorientation. Computed tomography revealed de novo SDH at the cerebellar tentorium. Computed tomography angiography and magnetic resonance imaging demonstrated TICA in the PCA. The patient was diagnosed with SDH due to a ruptured PCA aneurysm at the quadrigeminal segment. To avoid SDH growth due to re-rupture of the aneurysm, parent artery occlusion was subsequently performed with no complications. The patient was discharged home 2 months after endovascular treatment, with moderate disability. Follow-up angiography 2 years after the operation showed no recanalization, and the patient had returned to work.
CONCLUSIONS: TICA in the PCA can cause tentorial SDH with or without the presence of subarachnoid hemorrhage. Routine cerebrovascular assessment is crucial for head trauma with hematoma adjacent to the cerebellar tentorium. Parent artery occlusion via an endovascular procedure is an alternative treatment for TICA in the PCA that is less invasive than other approaches.
Keywords: Brain Injuries, Traumatic, Embolization, Therapeutic, Hematoma, Subdural, Acute, intracranial aneurysm, Magnetic Resonance Imaging, Subarachnoid Hemorrhage, Aneurysm, Ruptured, Cerebral Angiography, endovascular procedures, Hematoma, Subdural, Humans, Male
Background
The prevalence of nontraumatic ruptured intracranial aneurysm associated with subdural hematoma (SDH) without subarachnoid hemorrhage (SAH) or intraparenchymal hemorrhage is extremely low [1,2]. SDH can also be caused by traumatic intracranial aneurysm (TICA) [3]; however, diagnostic and therapeutic methods have not been established for clinical settings because TICA account for less than 1% of all cerebral aneurysms [4]. TICAs are known to rupture easily, resulting in a high mortality rate [5]. Accurate diagnosis and treatment are thus crucial for preserving life. Here, we describe a case of traumatic delayed SDH caused by a posterior cerebral artery (PCA) aneurysm, and we review the relevant literature.
Case Report
ENDOVASCULAR PROCEDURE:
Endovascular treatment was performed under general anesthesia. A guiding sheath (Flexor Shuttle; Cook Medical, Inc., Bloomington, IN, USA) was inserted into the right femoral artery and guided into the left vertebral artery. An intermediate catheter (TACTICS; Technocrat Corporation, Aichi, Japan) was guided into the left vertebral artery. A microcatheter (Excelsior SL-10; Stryker, Kalamazoo, MI, USA) was guided into the PCA on the proximal side of the pseudoaneurysm using a microwire (Synchro2; Stryker). PAO was performed using 5 coils (Target; Stryker) (Figure 3B, 3C).
POSTOPERATIVE CLINICAL COURSE:
Postoperatively, the patient was administered antiplatelet agents to prevent cerebral infarction, vasodilators to control blood pressure, and anticonvulsants to prevent symptomatic epilepsy. He was also given adequate saline to prevent dehydration. No cerebral infarction, rebleeding, or seizure was observed. Follow-up left VAG performed 2 weeks after embolization demonstrated complete occlusion with no recanalization. The patient was discharged home 2 months after endovascular treatment, with a Glasgow Outcome Scale (GOS) score of 4. Follow-up left VAG performed 2 years after the operation showed no recanalization (Figure 3D), and the patient had returned to work (GOS score of 5).
Discussion
TICA is found in less than 1% of intracranial aneurysms [4], and it may be misdiagnosed following head injury causing SDH. TICA is associated with significant morbidity and mortality rates, as high as 50% [5]; therefore, early diagnosis and treatment are crucial. TICA is generally due to severe traumatic brain injury (TBI) [6]. Most such aneurysms occur near the skull base or falx cerebri. The pathogenesis of TICA associated with head trauma is direct vascular injury or arterial extension by adjacent forces [5]. Vascular injury results in dissection with intramural hematoma or vessel outpouching, causing true, false, or mixed-type aneurysms [7]. Based on these mechanisms, if hematoma is found near the skull base or in parafalcine structures on non-contrast CT after severe TBI, further investigation should be attempted to explore the possibility of TICA [7]. In a review of 171 cases of TICA reported since 1960, Komiyama et al found that the anterior circulation is the most common site of TICA. Among these cases, a high frequency of occurrence was seen in the distal anterior cerebral artery (25%), cavernous portion of the internal carotid artery (ICA) (20%), distal middle cerebral artery (19%), and intradural portion of the ICA (14%) [8]. In contrast, only 8% of TICA cases reportedly involve posterior circulation sites [8].
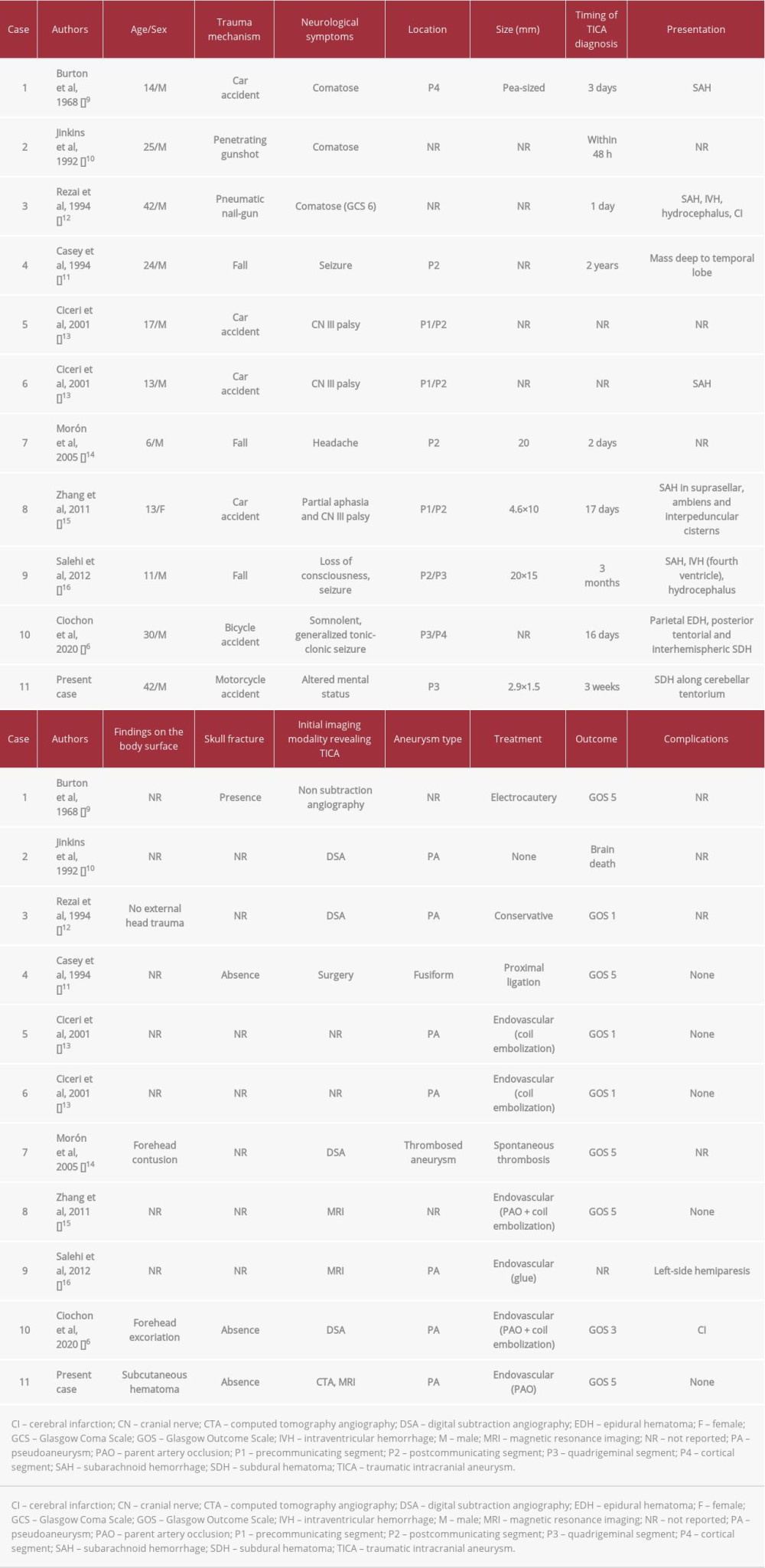
A summary of the literature regarding patients with TICA in the PCA is shown in Table 1 [6,9–16]. The mean age of patients was 19.5±10.8 years. Most aneurysms develop following traffic accidents, suggesting that this pathology most commonly involves younger individuals, or possibly that most older individuals who develop this pathology following traffic accidents die before it can be diagnosed. Typical symptoms include altered mental status, seizure, cranial nerve III palsy, headache, and partial aphasia. Most aneurysms were located in the precommunicating segment (P1) and postcommunicating segment (P2) of the PCA. Ruptured aneurysm resulting in only SDH, without SAH, is quite rare in the posterior fossa [17], and we identified only 1 case attributable to ruptured TICA involving the PCA [6]. The generally accepted mechanisms by which a ruptured cerebral aneurysm results in SDH are as follows: (i) continuous small hemorrhages from an aneurysm causes adhesions that eventually rupture in the subdural space; (ii) the systolic pressure generated by a ruptured aneurysm can cause the adjacent arachnoid membrane to rupture locally, spilling both cerebrospinal fluid and blood into the subdural space; and (iii) massive bleeding causes laceration of the cortex and arachnoid membrane [18]. In the present case, judging from the existence of SAH from the initial head trauma, the arachnoid membrane and PCA had been damaged at the cerebellar tentorium. Consequently, an aneurysm developed at the same site, and ruptured from the aneurysmal dome protruding into the subdural space, resulting in delayed SDH. Ruptured TICA in PCA should be suspected when delayed SDH is found at the cerebellar tentorium during conservative follow-up for traumatic intracranial hemorrhage.
In most cases, TICA has been diagnosed by non-subtraction or subtraction angiography (ie, DSA), not CTA (Table 1). Traditionally, DSA is the criterion standard test for detecting aneurysms [19]. Teksam et al pointed out that CTA could not take the place of DSA for detecting intracranial aneurysms, despite the developments in imaging modalities over the past few years as non-invasive options [19]. Further, Ciochon et al reported that pseudoaneurysm appearing as a large, left-sided tentorial and falcine SDH with a contrast extravasation following CTA, termed the spot sign, was initially interpreted as blood of varying age, resulting in delayed discovery of the pseudoaneurysm [6]. CTA alone may not be sufficient to delineate TICA. On the other hand, DSA has several shortcomings, including invasiveness due to the arterial puncture and intra-arterial catheter procedure, the need for expert proficiency with the procedure, and its comparatively high expense [19]. Regarding infectious intracranial aneurysm, some authors have already concluded that MRI is useful for diagnosing microaneurysms that have developed in the peripheral vasculature [20–23]. Cho et al point out that suggestive MRI findings can offer a sensitive and specific tool for identifying infectious intracranial aneurysm [24]. Similarly, such findings can be useful for the diagnosis and identification of TICA because TICA, like infectious intracranial aneurysm, usually occurs in peripheral blood vessels. For detecting TICA, MRI used together with CTA could provide accurate diagnosis. In the present case, because of the small size of the pseudoaneurysm, with a diameter less than 3 mm, diagnosis could not be achieved from CTA alone. Visualization of the pseudoaneurysm was enhanced by conducting MRI, with the combined results eventually leading to reliable diagnosis.
TICA can cause bleeding hours to weeks or even years after the trauma [25]. Komiyama et al noted that, especially when early signs of wall irregularity, narrowing, and spasm of the intracranial vasculature are detected on the initial angiogram, repeat DSA 1 week after TBI is recommended to rule out vascular injury [8]. In the present case, no vascular examination was performed after the injury. Whether the aneurysm appeared at the time of the TBI or a few days later has not been determined. Regarding TICA in the PCA, most aneurysms are identified within 3 weeks after head injury, but more than 3 months are required in some cases (Table 1). Therefore, follow-up imaging should be performed for at least 3 months. Routine cerebrovascular assessment, adding MRI data to the CTA data, is crucial in cases of severe head trauma with hematoma at the skull base or adjacent to the cerebellar tentorium.
TICA shows a high mortality rate with conservative treatment [5,7]. Rezai et al experienced a case of TICA in PCA treated conservatively with a GOS 1 outcome [12]. Craniotomy with clipping, resection, or trapping of the aneurysm was previously the first priority, but endovascular surgery has become the main treatment option, especially for PCA aneurysms [6,13,15,16], because of the reduced invasiveness of the procedure. Endovascular coil embolization into the pseudoaneurysm is not an appropriate treatment due to the high risk of intraoperative rupture and postoperative recanalization [7,26]. When Ciceri et al performed PAO on 7 PCA aneurysms, 1 patient developed contralateral hemiparesis and homonymous hemianopsia, and 1 patient presented with homonymous hemianopsia. In both cases, the aneurysm was in the P2/3 junction. Because the perforating arteries to the brainstem branch off from the P1 or P2 segments, PAO in this region is associated with brainstem infarction. In contrast, no complications occurred with PAO for peripheral vessels from P3 because the area of the cortical branches of the PCA has abundant collateral circulation with other arteries [13]. Based on the above, PAO for vessels distal to P3 is acceptable. Endovascular trapping is the best treatment in terms of preventing retrograde collateral filling from other vessels [27]. However, aneurysms occur in peripheral blood vessels, making it difficult to guide a microcatheter into the distal part of the aneurysm for trapping [7]. In the present case, because the pseudoaneurysm was in the P3 of the PCA, PAO was conducted with curative intent. Postoperatively, no cerebral infarction or re-bleeding was observed. In the future, treatment with new endovascular devices such as stents and flow diverters may make it easier to treat TICA without occluding the parent vessel [28].
Conclusions
TICA in the PCA can cause tentorial SDH. SAH may or may not be present. Routine cerebrovascular assessment should be performed for head trauma with hematoma adjacent to the cerebellar tentorium. PAO via an endovascular approach is a treatment option for TICA in the P3 of the PCA because of its reduced invasiveness, in addition to safety and efficacy of the procedure, compared with other surgical options, including craniotomy and intra-aneurysmal embolization.
Figures
References:
1.. Gong J, Sun H, Shi XY, Pure subdural haematoma caused by rupture of middle cerebral artery aneurysm: Case report and literature review: J Int Med Res, 2014; 42; 870-78
2.. Mrfka M, Pistracher K, Augustin M, Acute subdural hematoma without subarachnoid hemorrhage or intraparenchymal hematoma caused by rupture of a posterior communicating artery aneurysm: Case report and review of the literature: J Emerg Med, 2013; 44; e369-73
3.. Murakami M, Kakita K, Hosokawa Y, Ruptured traumatic aneurysm after trivial injury mimicking acute spontaneous subdural hematoma – case report: Neurol Med Chir (Tokyo), 2003; 43; 130-33
4.. Holmes B, Harbaugh RE, Traumatic intracranial aneurysms: A contemporary review: J Trauma, 1993; 35; 855-60
5.. Larson PS, Reisner A, Morassutti DJ, Traumatic intracranial aneurysms: Neurosurg Focus, 2000; 8; e4
6.. Ciochon UM, Steuble Brandt EG, Stavngaard T, Acute tentorial subdural hematoma caused by rupture of the posterior cerebral artery after minor trauma – a case report: Diagnostics (Basel), 2020; 10(3); 175
7.. Kumar A, Jakubovic R, Yang V, Traumatic anterior cerebral artery aneurysms and management options in the endovascular era: J Clin Neurosci, 2016; 25; 90-95
8.. Komiyama M, Morikawa T, Nakajima H, “Early” apoplexy due to traumatic intracranial aneurysm – case report: Neurol Med Chir (Tokyo), 2001; 41; 264-70
9.. Burton C, Velasco F, Dorman J, Traumatic aneurysm of a peripheral cerebral artery. Review and case report: J Neurosurg, 1968; 28; 468-74
10.. Jinkins JR, Dadsetan MR, Sener RN, Value of acute-phase angiography in the detection of vascular injuries caused by gunshot wounds to the head: analysis of 12 cases: Am J Roentgenol, 1992; 159; 365-68
11.. Casey AT, Moore AJ, A traumatic giant posterior cerebral artery aneurysm mimicking a tentorial edge meningioma: Br J Neurosurg, 1994; 8; 97-99
12.. Rezai AR, Lee M, Kite C, Traumatic posterior cerebral artery aneurysm secondary to an intracranial nail: Case report: Surg Neurol, 1994; 42; 312-15
13.. Ciceri EF, Klucznik RP, Grossman RG, Aneurysms of the posterior cerebral artery: Classification and endovascular treatment: Am J Neuroradiol, 2001; 22; 27-34
14.. Morón F, Benndorf G, Akpek S, Spontaneous thrombosis of a traumatic posterior cerebral artery aneurysm in a child: Am J Neuroradiol, 2005; 26; 58-60
15.. Zhang C, Chen H, Bai R, Traumatic aneurysm on the posterior cerebral artery following blunt trauma in a 14-year-old girl: Case report: Neuropediatrics, 2011; 42; 204-6
16.. Salehi MG, Ghanaati H, Abedini M, Traumatic dissecting posterior cerebral artery aneurysm. A case report and review of the literature: Neuroradiol J, 2012; 25; 563-68
17.. Missori P, Fenga L, Maraglino C, Spontaneous acute subdural hematomas. A clinical comparison with traumatic acute subdural hematomas: Acta Neurochir (Wien), 2000; 142; 697-701
18.. Gelabert-Gonzalez M, Iglesias-Pais M, Fernández-Villa J, Acute subdural haematoma due to ruptured intracranial aneurysms: Neurosurg Rev, 2004; 27; 259-62
19.. Teksam M, McKinney A, Casey S, Multi-section CT angiography for detection of cerebral aneurysms: Am J Neuroradiol, 2004; 25; 1485-92
20.. Subramaniam S, Puetz V, Dzialowski I, Cerebral microhemorrhages in a patient with mycotic aneurysm: Relevance of T2-GRE imaging in SBE: Neurology, 2006; 67; 1697
21.. Klein I, Iung B, Wolff M, Silent T2* cerebral microbleeds: A potential new imaging clue in infective endocarditis: Neurology, 2007; 68; 2043
22.. Morofuji Y, Morikawa M, Yohei T, Significance of the T2*-weighted gradient echo brain imaging in patients with infective endocarditis: Clin Neurol Neurosurg, 2010; 112; 436-40
23.. Fujimoto T, Morofuji Y, Matsunaga Y, Early diagnosis of infective endocarditis by brain T2*-weighted magnetic resonance imaging: Circ J, 2018; 82; 464-68
24.. Cho SM, Marquardt RJ, Rice CJ, Cerebral microbleeds predict infectious intracranial aneurysm in infective endocarditis: Eur J Neurol, 2018; 25; 970-75
25.. Chedid MK, Vender JR, Harrison SJ, Delayed appearance of a traumatic intracranial aneurysm. Case report and review of the literature: J Neurosurg, 2001; 94; 637-41
26.. Yuen CM, Kuo YL, Ho JT, Rapid regrowth of a successfully coiled traumatic pericallosal aneurysm: J Clin Neurosci, 2007; 14; 1215-19
27.. van Rooij WJ, Sluzewski M, Beute GN, Endovascular treatment of posterior cerebral artery aneurysms: AJNR Am J Neuroradiol, 2006; 27; 300-5
28.. Cohen JE, Gomori JM, Segal R, Results of endovascular treatment of traumatic intracranial aneurysms: Neurosurgery, 2008; 63; 476-85
Figures
In Press
12 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943244
13 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943275
13 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943411
13 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942864
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250