08 December 2021: Articles 
Stinging Nettle (): An Unusual Case of Galactorrhea
Unusual or unexpected effect of treatment, Adverse events of drug therapy
Laura Easton12DEF, Shalini Vaid34ACDEF, Angela K. Nagel5BDEF, Jineane V. Venci6DEF, Robert J. Fortuna126ABDEF*DOI: 10.12659/AJCR.933999
Am J Case Rep 2021; 22:e933999
Abstract
BACKGROUND: The increasing popularity and availability of herbal supplements among patients necessitates a better understanding of their mechanism of action and the effects they have on the body, both intended and unintended. Stinging nettle (Urtica dioica) is an herbaceous shrub found throughout the world that has been used for medicinal purposes for centuries.
CASE REPORT: A 30-year-old woman with obesity and GERD presented to a primary care clinic with new-onset galactorrhea. A urine pregnancy test was negative. Prolactin, thyroid-stimulating hormone (TSH), and a metabolic panel were all within normal limits. A mammogram demonstrated scattered areas of fibroglandular density and benign-appearing calcifications in the left breast. The breast ultrasound showed no suspicious findings. Her medications included intermittent Echinacea, etonogestrel implant 68 mg subdermal, and the supplement stinging nettle 500 mg, which she had been taking over the past month for environmental allergies. After consultation with a clinical pharmacist, the stinging nettle was discontinued. No additional changes to her medications or supplements were made. One week after discontinuation, she returned to the clinic with complete resolution of the galactorrhea.
CONCLUSIONS: Stinging nettle (Urtica dioica) is a common supplement and has effects on (1) sex hormone-binding globulin, (2) histamine-induced prolactin release, and (3) serotonin-induced release of thyrotropin-releasing hormone. The local estrogen bioactivity in breast tissue may subsequently lead to gynecomastia and/or galactorrhea. Supplements are an often overlooked but a critical component of medication reconciliation and potential clinical adverse effects.
Keywords: Dietary Supplements, Galactorrhea, herbal, Urtica dioica, Amenorrhea, Female, Humans, Male, Pregnancy
Background
Stinging nettle (
Case Report
A 30-year-old woman with a past medical history significant for obesity, GERD, and several abnormal pap smears presented to a primary care clinic with new galactorrhea. Three days prior to presentation, she developed intermittent white discharge from both breasts. The discharge occurred in the morning after showering and appeared like colostrum, similar to that seen in a prior pregnancy. At that time, she was seen in an urgent care facility and was prescribed antibiotics for a presumed breast infection, but she never received or took the antibiotic.
On the day of presentation, the patient was evaluated in person and a detailed history was obtained. She had no concerns for pregnancy. She had an etonogestrel implant in place for the past 1.5 years and she had not been pregnant or been breast-feeding in the past 2 years. Additional history was negative for any abnormal menstrual bleeding, skin changes, breast masses, or history of breast stimulation. Her medications included an etonogestrel implant placed 1.5 years ago, intermittent Echinacea, and stinging nettle 500 mg, which she started taking approximately 1 month prior for environmental allergies. Other than the introduction of the stinging nettle, there had been no other recent changes to her medications or supplement use. Review of systems was only notable for weight gain in the past few months. She denied any headaches, vision changes, abdominal pain, nausea, or vomiting. She denied any tobacco use, alcohol use, or illicit drugs.
A physical exam was notable for an anxious, tearful patient. Vital signs were within normal limits and BMI was 42 kg/m2.
Her breast exam demonstrated fibrocystic changes in both breasts and a small 0.5-cm, firm, non-mobile nodule in the right breast in the 4: 00 position. There were no abnormalities of the skin overlying the breast or the nipples. A neurologic exam demonstrated normal strength, symmetric sensation, and intact visual fields.
A urine pregnancy test was negative. Prolactin, thyroid-stimulating hormone (TSH), and a metabolic panel were obtained, and all were within normal limits. Given the nodule felt on the initial exam, she was sent for mammogram and targeted bilateral breast ultrasound. The mammogram showed scattered areas of fibroglandular density and benign-appearing calcifications in the left breast. The ultrasound showed no suspicious findings.
Etonogestrel and stinging nettle were both considered as possible causative agents of the galactorrhea [3]. Etonogestrel has galactorrhea listed in the package insert as a rare post-marketing reaction [4]. The etonogestrel implant was not removed based on the patient’s preference, long-term stability with the implant, and continued desire for contraception. The patient continued Echinacea since she had tolerated this supplement for over a year. After consultation with a clinical pharmacist, the supplement stinging nettle alone was discontinued. One week after discontinuation, she returned to the clinic with complete resolution of the galactorrhea.
Discussion
The differential diagnosis for galactorrhea is broad, including pregnancy, breast stimulation, hyperprolactinemia, thyroid disease, medications, or malignancy [5]. Multiple medications that block dopamine receptors (eg, phenothiazines, metoclopramide, tricyclic antidepressants), deplete dopamine (eg, methyldopa), inhibit dopamine release (eg, morphine), or block histamine receptors (eg, cimetidine) have been associated with galactorrhea. Etonogestrel has galactorrhea listed in the package insert as a rare post-marketing reaction, but this is rarely clinically observed [4]. Similarly, Echinacea does not have known associations with galactorrhea. After a thorough work-up, the patient’s galactorrhea was attributed to the supplement stinging nettle (
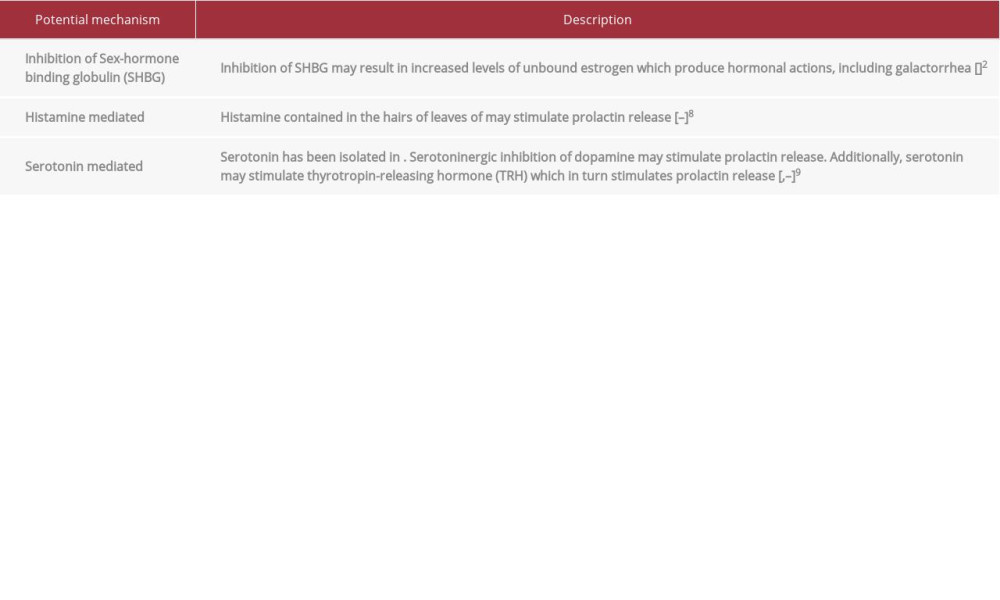
Although the mechanism is not completely understood, there are several potential mechanisms postulated for
In addition to directly binding to sex hormone-binding globulin,
Supplements are an overlooked but critical component of medication reconciliation. Patients often misperceive herbal products as natural and low risk and may neglect to mention their use to health care providers [15]. Thus, it is important that patients are asked specifically about supplement use during medication reconciliation and in evaluation for suspected drug-related problems.
Unlike prescription and over-the-counter medications, the Food and Drug Administration does not require herbal supplements to undergo rigorous safety and efficacy testing prior to sales. This paucity of literature complicates proactive identification of adverse effects and potential interactions. Incidences of harm are thus collected most often through case reports. Accordingly, health care providers should remain vigilant about reporting adverse effects to the Food and Drug Administration, through MedWatch Online Voluntary Reporting Form (https://www.safetyreporting.hhs.gov). To maintain the provider-patient relationship, providers must balance respecting patient beliefs regarding supplement use with the best available evidence regarding safety and efficacy. To the best of their ability, providers should provide patients with guidance regarding safe use of herbal products. We find that the Natural Medications database is the most comprehensive source of such data [16].
Conclusions
Stinging nettle (
References:
1.. Lust JB: The Herb Book: The Most Complete Catalog of Herbs Ever Published, 2014; 1-617, Mineola, New York, Dover Publications, Inc
2.. , Monograph: Altern Med Rev, 2007; 12(3); 280-84
3.. Visconti F, Zullo F, Marra ML, A new long-term reversible contraception method: Sexual and metabolic impact: Transl Med UniSa, 2012; 4; 86-89
4.. Co M.: Nexplanon [package insert], 2015, NJ, Whitehouse Station
5.. Jameson JL: Harrison’s Principles of Internal Medicine, 2020, McGraw Hill
6.. Zapantis A, Steinberg JG, Schilit L, Use of herbals as galactagogues: J Pharm Pract, 2012; 25(2); 222-31
7.. Sahin M, Yilmaz H, Gursoy A: N Z Med J, 2007; 120(1265); U2803
8.. Emmelin N, Feldberg W, The mechanism of the sting of the common nettle (urtica urens): J Physiol, 1947; 106(4); 440-55
9.. Upton R: Journal of Herbal Medicine, 2013; 3(1); 9-38
10.. Müller EE, Locatelli V, Cella S, Prolactin-lowering and -releasing drugs. Mechanisms of action and therapeutic applications: Drugs, 1983; 25(4); 399-432
11.. Collier HO, Chesher GB: Br J Pharmacol Chemother, 1956; 11(2); 186-89
12.. Aggarwal A, Kumar R, Sharma RC, Escitalopram induced galactorrhoea: A case report: Prog Neuropsychopharmacol Biol Psychiatry, 2010; 34(3); 557-58
13.. Peterson MC, Reversible galactorrhea and prolactin elevation related to fluoxetine use: Mayo Clin Proc, 2001; 76(2); 215-16
14.. Sternbach H, Venlafaxine-induced galactorrhea: J Clin Psychopharmacol, 2003; 23(1); 109-10
15.. Lynch N, Berry D, Differences in perceived risks and benefits of herbal, over-the-counter conventional, and prescribed conventional, medicines, and the implications of this for the safe and effective use of herbal products: Complement Ther Med, 2007; 15(2); 84-91
16.. , 2021 9/14/2021]; Available from: https://naturalmedicines.therapeuticresearch.com/
17.. , “Stinging Nettle stinging bits” by John Tann is licensed with CC BY 2.0 To view a copy of this license, visithttps://creativecommons.org/licenses/by/2.0/
In Press
06 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942937
12 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943244
13 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943275
13 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943411
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250