14 October 2020: Articles 
Limitations of Dutch Growth Research Foundation Commercial Software Weight Velocity for Age Standard Deviation Score
Rare disease
Martin J.C. Van Gemert1ABCDEF*, Marianne Vlaming2ABDEF, Bülent Köseoğlu3ABCD, Cornelis M.A. Bruijninckx4ABCDE, Ton G. Van Leeuwen1ACDE, Martino H.A. Neumann5ACDEF, Pieter J.J. Sauer6ABCDEDOI: 10.12659/AJCR.925551
Am J Case Rep 2020; 21:e925551
Abstract
BACKGROUND: The commercial software for hospitals, Weight Velocity for Age Standard Deviation Score (SDSWVA), claims to document the growth and development of children, although published details are unavailable. The statistics-derived parameter SDSWVA includes the weight velocity at age t, WV(t) (weight gained between t and (t–1.23) years, divided by 1.23), and 3 standard weight velocity curves at average age AA, defined as AA=t–1.23/2 years. SDSWVA denotes the number of standard deviations that WV(t) deviates from the 0 SD weight velocity at AA. WV(t) yielded erroneous outcomes when applied to weights of a seriously underweight boy with an allergy to cows’ milk who showed strong weight growth after being fed on food free of cows’ milk. The SDSWVA software tacitly suggests that it is more accurate than WV(t).
CASE REPORT: The case of this boy was previously described in this Journal. Using SDSWVA(t,AA) software, his weight growth was analyzed by his third pediatrician, beginning at age 1.5 years. The diagnosis of the mother with Pediatric Condition Falsification was confirmed, adding 6 months to foster care, which totalled 8.5 months. Testing of the SDSWVA software on the boy’s weight curve yielded results that were complex, nontransparent, and as erroneous as WV(t), explaining the misdiagnosis by the third pediatrician.
CONCLUSIONS: SDSWVA software should not be used for children under 3 years and during variable weight behavior. Erroneous performance, unpublished details, and an error identified in their new but untested software make the Dutch Growth Research Foundation unlikely to meet the 2020 European Union regulations for in vitro medical devices.
Keywords: Body Weight Changes, case reports, Diagnostic Errors, Software, Child, Child, Preschool, Infant
Background
The
We previously showed that
In the Appendix below we explain how the
Case Report
EARLIER:
The erroneous behavior of WV(t) was evident when applied to the weight curve of an infant boy [1]. Figure 1 shows his weight curve at 15 consecutive age periods (see Schematic Model below). Briefly [3], the boy was born at 39 gestational weeks as the sixth child of normal parents, weighting 3.18 kg. He was hospitalized for 2 weeks during age period 2 because of a slightly negative weight gain. Allergy to cows’ milk was suspected, with subsequent removal of cows’ milk from his diet resulted in a rapid weight gain (periods 3 and 4). Despite impressive weight growth, during periods 3–11 (age 0.33–2.4 years), which was 1.3- to 2.3-fold greater than the corresponding weight growth on the 0 SD standard weight curve, his first pediatrician stated in a legal summary of the second of 3 juvenile court hearings held in the boy’s case that “the boy does not grow” and ordered his mother to increase his food intake stepwise to 3.5 times normal (period 8) [3]. During period 8 (period 6 of [3]), the boy’s weight velocity was 2.1 times the 0 SD weight velocity. This pediatrician, as well as the second pediatrician, who was willing to confirm all the erroneous statements made by the first pediatrician during the second juvenile court hearing, appeared unable to distinguish (low) weight from (exceptional) weight growth [3]. Based on these reports, the mother was diagnosed with PCF and the boy was placed in foster care for 8.5 months.
CASE REPORT:
The present case begins during period 10, after the boy was in foster care for 2.5 months, at the time his third pediatrician was appointed. This pediatrician analyzed his weight growth with
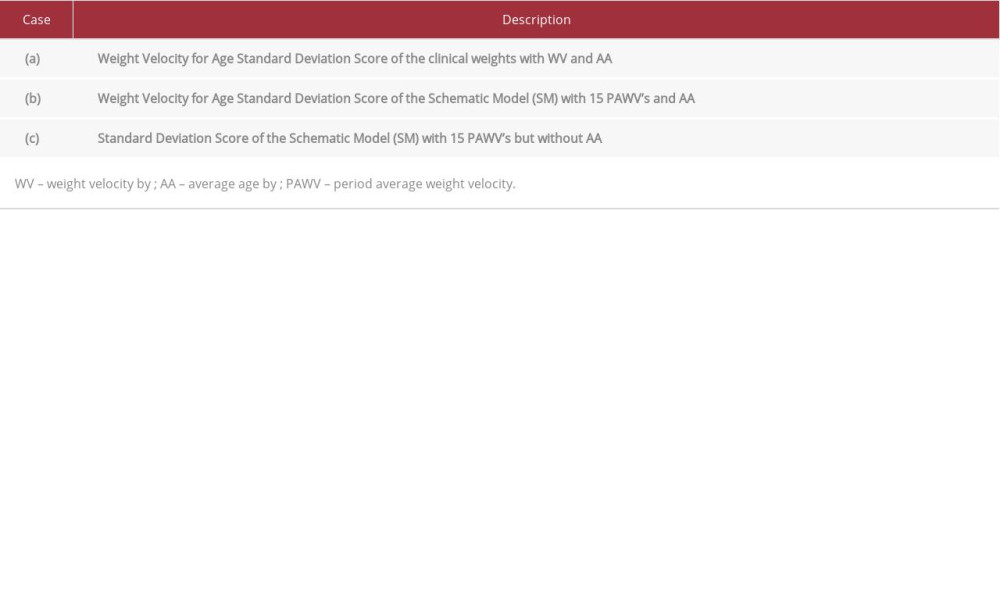
SDS(t) and SDSSM(t,AA) were applied to 2 weight curves of this boy (Figure 1). The first weight curve (the clinical weights) was of the actual measured weights of the boy until age 3.1 years [1,3], whereas the second weight curve, the Schematic Model of his weights [1], replaced the individually measured weights with weights clustered in 15 consecutive age periods by least-squares fitting. The virtually linear increase in weight in all age periods gave 15 individual but accurate Period-Averaged-Weight-Velocities (Table 1 in [1], summarized in the caption to Figure 1). For comparison we also show the 0 SD standard weight curve.
We calculated (a) SDSWVA(t,AA), the Weight Velocity for Age Standard Deviation Score of the boy’s clinical weights, and (b) SDSSM(t,AA), the Weight Velocity for Age Standard Deviation Score of the Schematic Model [1] with their exact Period-Averaged-Weight-Velocities, but including average age AA. We compared these 2 predictions with (c) SDSSM(t), the exact Standard Deviation Score of the Schematic Model with Period-Averaged-Weight-Velocities but without AA, here considered the standard for SDS-calculations, as these are arguably the most exact approximations of real weight growth velocity. The Table 1 summarizes the 3 case examples. This approach shows the effects on SDS-calculations of natural weight fluctuations, the 1.23 years of inter-weight age interval for WV, and the use of an average age AA.
Results
Figure 1 shows that, when the boy’s life became normal again, the
Discussion
This study showed that the software package of the
The key finding of this study was that the
Erroneous outcomes of
Additionally, we purchased version
Conclusions
The
Figures
References:
1.. van Gemert MJC, Bruijninckx CMA, van Leeuwen TG, Limitations of weight velocity analysis by commercial computer program Growth Analyser Viewer Edition: Ann Biomed Eng, 2019; 47(1); 297-305
2.. Ernst M, Bernhardt M, Bechstein M, Performance of semiautomatic assessment of carotid artery stenosis on CT angiography: Clarification of differences with manual assessment. Effect of CAD on performance in ASPECTS reading: Inform Med Unlocked, 2020; 18; 100295
3.. van Gemert MJC, Vlaming M, Osinga E, Pediatric condition falsification misdiagnosed by misjudged weight growth from the curve of measured weights: Am J Case Rep, 2018; 19; 752-56
4.. van Gemert MJC, Bruijninckx CMA, Neumann HAM, Weight velocity equations with 14–448 days time separated weights should not be used for infants under 3 years of age: Med Hypotheses, 2019; 129; 109234
5.. Gerver WJM, de Bruin R: Paediatric morphometrics, 2001, Maastricht, University Press Maastricht
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133