12 August 2021: Articles 
Hepatotoxicity Induced by Fluvastatin: A Reversible Acute Cholestatic Liver Injury
Unusual clinical course, Challenging differential diagnosis, Unusual or unexpected effect of treatment, Diagnostic / therapeutic accidents, Adverse events of drug therapy
Najah S. Alanazi1ADEF, Tahani S. Alenazi1ADEF, Khalidah A. Alenzi1ABDEF*DOI: 10.12659/AJCR.931418
Am J Case Rep 2021; 22:e931418
Abstract
BACKGROUND: Fluvastatin, a commonly prescribed statin, is indicated for treatment of hypercholesterolemia in persons at high risk for coronary, cerebrovascular, and peripheral artery disease. However, there have been rare reports of liver injury or renal failure associated with use of fluvastatin.
CASE REPORT: We describe the case of a 69-year-old Saudi man with a medical history of diabetes mellitus and hypercholesterolemia for 2 years, on metformin, gliclazide modified release, daily aspirin, and simvastatin. Fluvastatin 40 mg daily was administered instead of simvastatin for 7 weeks before the patient was admitted to the hospital with fatigue, weakness, abdominal pain, loss of appetite, vomiting, itching, and elevated liver enzymes. Discontinuation of fluvastatin and other combined therapies led to a decrease in liver enzymes. He was diagnosed with fluvastatin-induced cholestatic liver injury and acute kidney disease.
CONCLUSIONS: The Naranjo scale indicates a probable relationship between cholestatic liver injury and fluvastatin, as well as a possible relationship between cholestatic injury and gliclazide and metformin. In our case report, we describe the synergistic effect of several factors in contributing to liver injuries, such as age, long-term gliclazide intake, and fluvastatin. Accordingly, we recommend close monitoring of patients’ liver and kidney function, especially in the elderly and those with polypharmacy, while allowing sufficient time for the liver function to recover from a reversible reaction to fluvastatin.
Keywords: Chemical and Drug Induced Liver Injury, Cholestasis, fluvastatin, Gliclazide, Metformin, Hydroxymethylglutaryl-CoA Reductase Inhibitors, simvastatin
Background
Liver injury can be induced in several ways, including, often, by medications. More than 900 drugs may be associated with liver injury. Approximately 20–40% of all cases of fulminant hepatic failure are caused by drugs, with liver transplantation and death the outcomes of 75% of idiosyncratic drug reactions [1]. Drug-induced liver injury (DILI) has been the most common cause for withdrawal of drugs from the market during the past decade [2]. Antidiabetic, anticonvulsant, anti-hypertensive, nonsteroidal anti-inflammatory drugs, and statins are common inducers of cholestatic liver injury [3]. Adverse drug reactions (ADRs) include any harmful and unintended effect that results from the use of a medication in normal doses for diagnosis, prevention, or treatment [4].
The Council of International Organizations of Medical Sciences defines cholestatic injury as a patient having an alkaline phosphate level of more than 2 times the upper limit of normal (ULN) or in whom alanine aminotransferase (ALT)/alkaline phosphatase (ALP) <2.
A mixed hepatocellular cholestatic injury can be defined as the presence of ALT/ALP level of between 2 and 5 and hepatocellular injury as the presence of ALT >2×ULN or ALT/ALP >5 [3]. After the causative medication is stopped, cholestatic reactions need time to return to normal, because hepatocyte recovery and regeneration take place more rapidly than the return of cholangiocyte and bile secretion activity to normal levels [3].
Statins are among the most commonly used drugs worldwide, owing to their efficacy, and have been shown to have a good safety profile at therapeutic doses. This class of drugs is used to lower cholesterol levels [5]. The most common adverse effects associated with the use of statins are elevation of liver enzymes, myalgia, and rhabdomyolysis. DILI caused by statins is generally rare but can be severe [6]. Statins can induce myositis and myopathy and consequently lead to renal failure and liver injury. In animal studies, very high doses of statins may cause hepatic cell necrosis, but normal therapeutic doses of the drug have not been associated with liver injury [7].
According to the Uppsala Monitoring Center, ADRs reported with the use of fluvastatin include jaundice (14%), hyperbilirubinemia (6.1%), DILI (3.7%), liver injury (3.3%), hypertransaminasemia (1.4%), cholestatic jaundice (0.6%), increased blood creatine phosphokinase (36.7%), acute kidney injury (23.4%), and chronic kidney disease (1.3%) [8]. These ADR reports can be retrieved from the VigiAccess database through URL http://www.vigiaccess.org/.
Gliclazide, a widely used therapy for type 2 diabetes, is generally considered a first-lines medication for its efficacy and safety profile. Clinically, liver injury induced by sulfonylureas has been rare [9].
Metformin is an oral hypoglycemic agent commonly used to treat type 2 diabetes mellitus, and metformin-associated metabolic acidosis is one of its commonly recognized adverse effects. Clinically apparent hepatic injury from metformin is rare [10].
Case Report
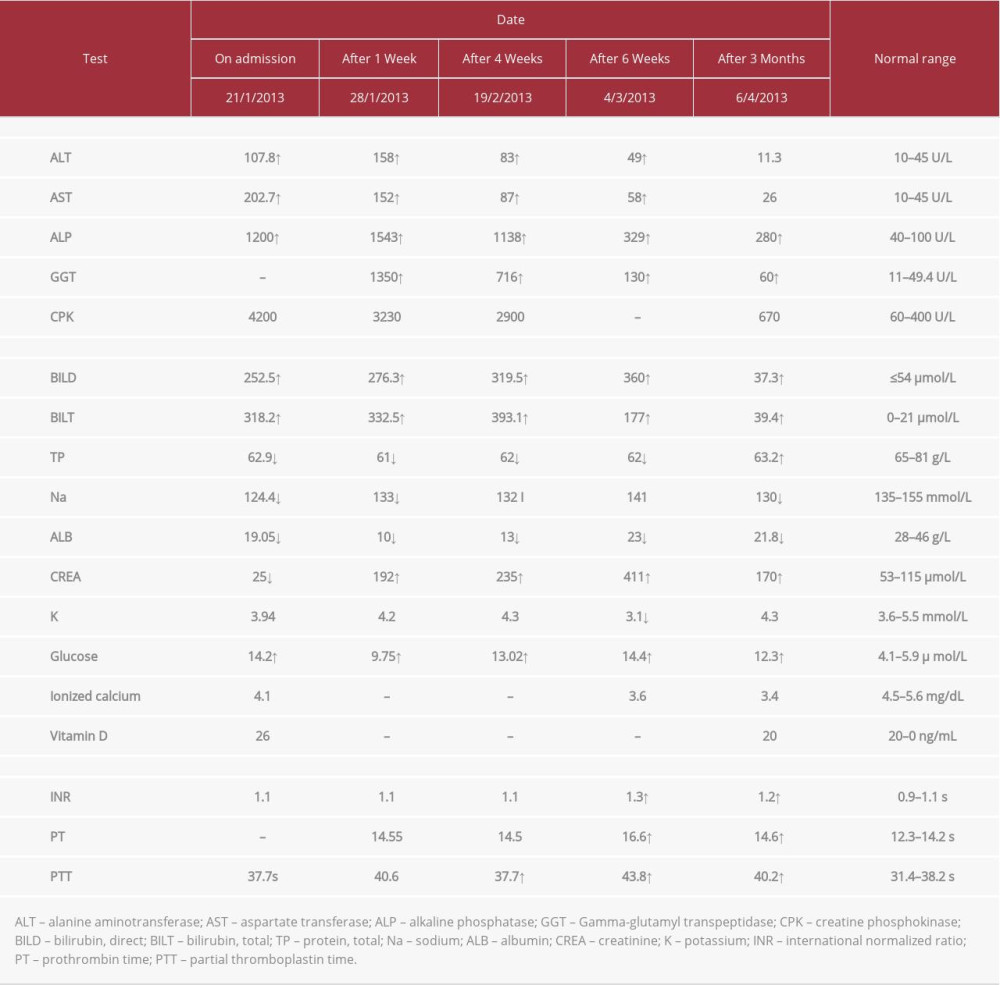
A 69-year-old man, a retired soldier with a medical history of diabetes mellitus and hypercholesterolemia for 2 years, was on metformin 500 mg 3 times a day, gliclazide modified release 30 mg daily, aspirin 81 mg daily, and simvastatin 20 mg, for 2 years. The patient’s physician replaced his simvastatin with fluvastatin 40 mg per day 7 weeks prior to the patient’s admission to the hospital. On admission, the patient presented with fatigue, weakness, abdominal pain, loss of appetite, vomiting, dark urine, and itching. Moreover, on examination, he was jaundiced and tender in the upper right quadrant of his abdomen, with yellowish discoloration of the skin. His vital signs were stable, and his heart and chest were normal, but he had a distended abdomen and an enlarged liver. Table 1 summarizes the laboratory findings.
An abdominal ultrasound report showed enlarged liver and no bile duct obstruction. Serology for hepatitis A, B, C, E, cytomegalo-virus (CMV), and Epstein-Barr virus (EBV) was negative. Serologic tests were negative for hepatitis B surface antigen, anti-hepatitis B core immunoglobulin M (IgM), anti-hepatitis A virus IgM, anti-hepatitis C virus antibody, anti-CMV IgM; anti-EBV IgM, antinuclear antibodies, anti-smooth-muscle antibodies, anti-mitochondrial antibodies, and anti-neutrophil cytoplasmic antibodies.
Fluvastatin, metformin, and gliclazide were discontinued on admission. One week later, laboratory results showed increased levels of AST, ALT, ALP, BILT, BILD, and creatinine.
After 4 weeks, he was referred to a specialist hospital because he appeared edematous. The patient was diagnosed with fluvastatin-induced cholestatic liver injury and acute renal failure, and was identified as a possible candidate for liver transplant. A liver biopsy was not performed on this patient.
We noticed from the last lab results that his creatinine level was increasing, and creatine phosphokinase was decreasing. The patient received supportive treatment at admission which included good hydration with normal saline. Additionally, the patient presented with mild hypocalcemia. Therefore, an oral supplement of calcium carbonate 500 mg twice daily was administered with alfacalcidol 1 mcg once daily. Also, a low-protein diet was followed by the patient. With supportive treatment, all hepatobiliary laboratory results completely recovered to normal levels. Kidney function stabilized at an estimated glomerular filtration rate (GFR) in the mid-30s, and the coagulation profile returned largely to normal. The patient has since been diagnosed with stage 3 chronic kidney disease (CKD); he is stable but requires routine follow-up.
Discussion
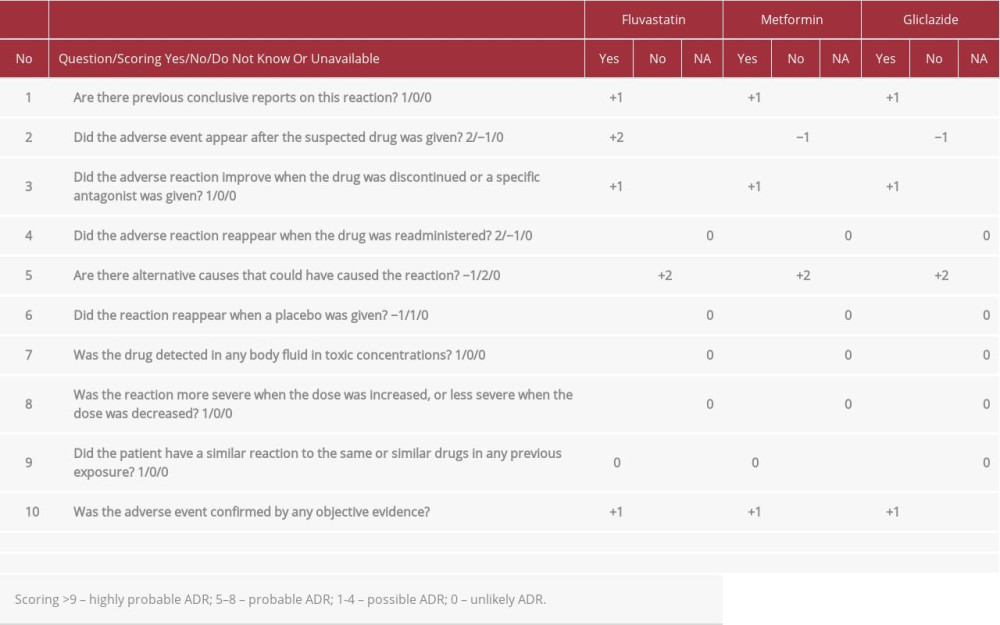
Our patient was diagnosed with cholestatic jaundice and myositis induced by fluvastatin, as indicated by high creatine phosphokinase level. Creatine phosphokinase elevation is the hallmark of rhabdomyolysis [11]. There have been cases in which fluvastatin was associated with ADRs such as myalgia, and with creatine phosphokinase elevation that led to discontinuation of therapy [12]. Creatine phosphokinase was decreasing due to recovery. The Naranjo algorithm is the most commonly used tool for assessing ADR causality. We applied this algorithm for liver injury and found causality ratings of 4 for gliclazide and metformin, indicating a possible ADR, and 7 for fluvastatin, indicating a probable ADR (Table 2) [13].
We also used the Maria and Victorino scale to examine the association between fluvastatin and cholestatic liver injury. This scale provides a scoring system involving 5 axes in the decision strategy based on chronological criteria, including (1) the temporal relationship between drug intake and the reaction (onset and withdrawal), as well as the course of the reaction; (2) exclusion of alternative causes such as viral hepatitis, alcoholic liver disease, or biliary obstruction; (3) extrahepatic manifestations (rash, fever, arthralgia, eosinophilia, cytopenia); (4) re-exposure to the drug (rechallenge); and (5) previous reports in the literature of cases of hepatotoxicity associated with the drug. Concordance with the 5 classic degrees of probability of adverse drug reactions is established based on the tabulated score as follows: “definite” (score >17), “probable” (score 14–17), “possible” (score 10–13), “unlikely” (score 6–9), and “excluded” (score <6) [14]. In the present case, gliclazide had a score of 14 points (possible), metformin, 14 points (possible), and fluvastatin, 16 points (possible), supporting a conclusion that fluvastatin, rather than metformin and gliclazide, could be the cause of injury.
The statins most commonly associated with DILI are atorvastatin and simvastatin, in that order. Based on total study cohort and in comparison with DILI from other drugs, fluvastatin had the highest proportional incidence of DILI, consistent with our results. However, a possible explanation for this might be the greater frequency of atorvastatin and simvastatin use compared with fluvastatin use, which hindered attempts to interpret this finding [5]. A meta-analysis found that higher doses of statins were associated with significant transaminase elevation [15].
The mechanism of hepatic injury induced by fluvastatin is still unknown, but fluvastatin is metabolized in the liver by the cytochrome P450 2C9 enzyme and excreted in bile. The idiosyncratic reaction can be explained by hypersensitivity, failure to adapt (a phenomenon marked by initial elevation in amino-transferase level, then a return to normal range), and elevation of ALT leading to toxic intermediates of fluvastatin metabolism. Other possible mechanisms are mitochondrial dysfunction, cell apoptosis, and the concomitant use of drugs that use the same metabolic pathway, such as cytochrome P450 2C9 enzymes (eg, gliclazide) [2].
Hepatotoxicity associated with sulfonylureas as an adverse effect is rare but has been reported [16]. The mechanism of liver injury caused by the sulfonylurea gliclazide is unknown but is suspected to result from hypersensitivity and immuno-allergic reaction [17].
Liver injury is rare with metformin, but the literature indicates a potential association between metformin and liver damage after administration and suggests careful long-term monitoring. Metformin is not metabolized in the liver, and possible mechanisms of acute hepatocellular or cholestatic jaundice may involve drug-drug interactions or direct or idiosyncratic reactions (hypersensitivity) [18–20].
The patient in this case developed reversible cholestatic liver injury, perhaps because fluvastatin was administered in a low-intensity dose, hepatotoxicity associated with the combined medications (metformin and gliclazide) is rare, the duration of administration was short, and there was no history of alcohol intake.
The patient had an abnormally high level of creatine kinase. It was probably for this reason that he developed rhabdomyolysis induced by fluvastatin, which consequently led to acute renal failure. Acute renal injury is the most serious complication of rhabdomyolysis, and it is diagnosed on the basis of elevated creatine kinase and myoglobin [21]. The present patient did not have elevated antineutrophil cytoplasmic antibodies, anti-glomerular basement membrane antibodies, positive antinuclear antibodies, or double-stranded DNA antibodies. Therefore, the diagnosis of acute interstitial nephritis caused by an immune-allergic reaction was ruled out, increasing the likelihood of acute kidney injury associated with rhabdomyolysis based on the high creatine kinase level [22].
The role of liver biopsy in the diagnosis of DILI is limited because there are no significant features that help to differentiate between DILI and another hepatic disease, in addition to the high complication rate associated with liver biopsy as an invasive procedure [23]. Therefore, liver biopsy was not performed and the patient was diagnosed with DILI based on the assessment of lab results and clinical examination.
The Roussel Uclaf Causality Assessment Method (RUCAM) was introduced in 1993 to assess the causality of medication-induced liver toxicity; it is now widely used for its ability to objectively evaluate the likelihood of a drug’s precipitating liver injury [24]. The RUCAM contains 7 criteria, as follows: onset time, clinical course, risk factors, other concomitant drugs used, published research on hepatotoxicity, reactions associated with re-administration of the drug, and non-drug-causes. The RUCAM score ranges from −8 to +14, with higher values indicating a greater degree of association. The RUCAM score found for fluvastatin’s probable association with a cholestatic pattern of liver injury indicated that the evidence supports a link between fluvastatin and liver injury. The ADR database sourced from the WHO and the Uppsala Monitoring Center revealed 917 hepatobiliary ADR reports on fluvastatin with variable hepatic disorders, about 61 reports on cholestatic liver injury, 34 confirmed reports on fluvastatin relating to drug-induced liver injury, 153 reports on metformin-induced liver injury, 10 reports on gliclazide-induced liver injury, and 82 reports on drug-associated liver function abnormality [8]. However, metformin does not undergo hepatic metabolism, and hepatotoxicity associated with it is rare; it has been documented only in cases with concomitant use of hepatotoxic drugs [19].
Gliclazide is a second-generation sulfonylurea with a documented risk of cholestatic jaundice due to its metabolism in the liver [25]. It was associated, in 2 case reports, with acute hepatitis and inflammation that resolved after discontinuation [26,16]. Because we could not exclude the possible contribution of metformin and gliclazide in the diagnosis of patients who had liver injury, we assigned −2 points in Step 4. We reported a cholestatic pattern of liver injury potentially associated with fluvastatin, based on RUCAM score. This result supports previous research from the Spanish Hepatotoxicity Registry that identified fluvastatin and atorvastatin as the most frequent causes of cholestasis among statins (66%) [27]. The probable causality of fluvastatin in liver injury may be explained by the dose-dependent toxicity of statins, with our patient’s medical history showing administration of simvastatin 20 mg followed by fluvastatin 40 mg. Similar outcomes were observed in a systemic review of 22 cases of statin-induced liver injury, in which fluvastatin was linked to probable causality of liver injury in 2 cases, both of which had a history of dosage increase more than a month before symptom onset [28].
The present case showed a transient elevation in liver enzymes 7 weeks after fluvastatin administration, followed by a rapid recovery 3 months after admission, much like in a prospective study of 8 cases that showed a transient and reversible increase in liver aminotransferase 2 weeks to 3 months after treatment with fluvastatin [29]. However, 1 case reported chronic liver injury after prolonged exposure to fluvastatin for more than 1 year. This different outcome may be explained by the short exposure to fluvastatin (7 weeks) in the present case compared with the prolonged latency of hepatotoxicity after long-term exposure seen in the case with chronic liver injury [28]. Moreover, our patient had no significant history of alcohol intake, minimizing the risk of liver damage and enhancing recovery. Because the prognosis for liver injury after fluvastatin intake varies by patient, careful monitoring of liver enzymes is recommended to avoid permanent liver injury.
Conclusions
The 3 scales that we used indicated an association between fluvastatin use and cholestatic liver injury and renal failure. The Naranjo scale in particular indicated a probable relationship between cholestatic liver injury and fluvastatin and a possible relationship between cholestatic liver injury and gliclazide and metformin. A combination of these therapies can induce cholestatic hepatic injury, but fluvastatin could be the most likely cause due to its higher score. This case reveals multiple factors contributing to liver injury, including age, long-term intake of gliclazide, and fluvastatin administration. We thus recommend careful monitoring of liver and renal function at baseline and periodically thereafter for patients on combined therapy (fluvastatin, metformin, and gliclazide), to minimize the hepatic adverse effects in the elderly. We also recommend a waiting period of approximately 2 months before deciding whether to perform a liver transplant, to allow for reversal in liver function in patients who have developed cholestatic liver injury after fluvastatin administration. Additionally, both healthcare professionals and the patient should be aware of this potential side effect.
References:
1.. Ostapowicz G, Fontana R, Schiodt F, Results of a Prospective Study of Acute Liver Failure at 17 Tertiary Care Centers in the United States: Ann Intern Med, 2002; 137(12); 947-54
2.. Karahalil B, Hare E, Koç G, Hepatotoxicity associated with statins: Arh Hig Rada Toksikol, 2017; 68(4); 254-60
3.. Padda MS, Sanchez M, Akhtar AJ, Boyer JL, Drug-induced cholestasis: Hepatology, 2011; 53(4); 1377-87
4.. Aronson J, Ferner R, Clarification of terminology in drug safety: Drug Saf, 2005; 28(10); 851-70
5.. Björnsson E, Jacobsen EI, Kalaitzakis E, Hepatotoxicity associated with statins: Reports of idiosyncratic liver injury post-marketing: J Hepatol, 2012; 56(2); 374-80
6.. Kshirsagar A, Vetal Y, Ashok P, Bhosle P, Ingawale D, Drug induced hepatotoxicity: A comprehensive review: Internet J Pharmacol, 2009; 8(2); 281668670
7.. Bhardwaj SS, Chalasani N, Lipid-lowering agents that cause drug-induced hepatotoxicity: Clin Liver Dis, 2007; 11(3); 597-613
8.. , VigiAccess database http://www.vigiaccess.org/
9.. Pandit A, Sachdeva T, Bafna P, Drug-induced hepatotoxicity: A review: J Appl Pharm Sci, 2012; 2(5); 233-43
10.. Gillett RC, Norrell A, Considerations for safe use of statins: liver enzyme abnormalities and muscle toxicity: Am Fam Physician, 2011; 83(6); 711-16
11.. Dimitriu A, Lupescu O, Ciurea N, Markers of inflammation in crushing trauma of the lower limbs: Ther Pharmacol Clin Toxicol, 2016; 20(1); 20-24
12.. Sessa M, Rafaniello C, Scavone C, Preventable statin adverse reactions and therapy discontinuation. What can we learn from the spontaneous reporting system?: Expert Opin Drug Saf, 2018; 17(5); 457-65
13.. Zaki SA, Adverse drug reaction and causality assessment scales: Lung India, 2011; 28(2); 152-53
14.. Maria VA, Victorino RM, Development and validation of a clinical scale for the diagnosis of drug-induced hepatitis: Hepatology, 1997; 26(3); 664-69
15.. Naci H, Brugts J, Ades T, Comparative tolerability and harms of individual statins: Circ Cardiovasc Qual Outcomes, 2013; 6(4); 390-99
16.. Dourakis SP, Tzemanakis E, Sinani C, Gliclazide-induced acute hepatitis: Eur J Gastroenterol Hepatol, 2000; 12(1); 119-21
17.. Russo MW, Scobey M, Bonkovsky HL, Drug-induced liver injury associated with statins: Semin Liver Dis, 2009; 29(4); 412-22
18.. Hashmi T, Probable hepatotoxicity associated with the use of metformin in type 2 diabetes: BMJ Case Rep, 2011; 2011; bcr0420114092
19.. Miralles-Linares F, Puerta-Fernandes S, Bernal-Lopez MR, Metformin-induced hepatotoxicity: Diabetes Care, 2012; 35(3); e21
20.. Shurrab NT, Arafa E-SA, Metformin: A review of its therapeutic efficacy and adverse effects: Obes Med, 2020; 17; 100186
21.. Al-Ismaili Z, Piccioni M, Zappitelli M, Rhabdomyolysis: Pathogenesis of renal injury and management: Pediatr Nephrol, 2011; 26(10); 1781-88
22.. Mercado M, Smith D, Guard E, Acute kidney injury: diagnosis and management: Am Fam Physician, 2019; 100(11); 687-94
23.. Teschke R, Frenzel C, Drug induced liver injury: Do we still need a routine liver biopsy for diagnosis today?: Ann Hepatol, 2014; 13(1); 121-26
24.. : LiverTox: Clinical research information on drug-induced liver Injury, 2012, Bethesda (MD), National Institute of Diabetes and Digestive and Kidney Diseases Roussel Uclaf Causality Assessment Method (RUCAM) in Drug Induced Liver Injury. [Updated 2019 May 4]. https://www.ncbi.nlm.nih.gov/books/NBK548272/
25.. Kalra S, Aamir AH, Raza A, Place of sulfonylureas in the management of type 2 diabetes mellitus in South Asia: A consensus statement: J Endocrinol Metab, 2015; 19(5); 577-96
26.. Chitturi S, Le V, Kench J, Gliclazide-induced acute hepatitis with hypersensitivity features: Dig Dis Sci, 2002; 47(5); 1107-10
27.. Perdices EV, Medina-Cáliz I, Hernando S, Hepatotoxicity associated with statin use: Analysis of the cases included in the Spanish Hepatotoxicity Registry: Rev Esp Enferm Dig, 2014; 106(4); 246-54
28.. Russo MW, Hoofnagle JH, Gu J, Spectrum of statin hepatotoxicity: Experience of the drug-induced liver injury network: Hepatology, 2014; 60(2); 679-86
29.. Guo M, Zhao J, Zhai Y, A prospective study of hepatic safety of statins used in very elderly patients: BMC Geriatr, 2019; 19; 352
In Press
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942826
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942770
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943214
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943010
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250